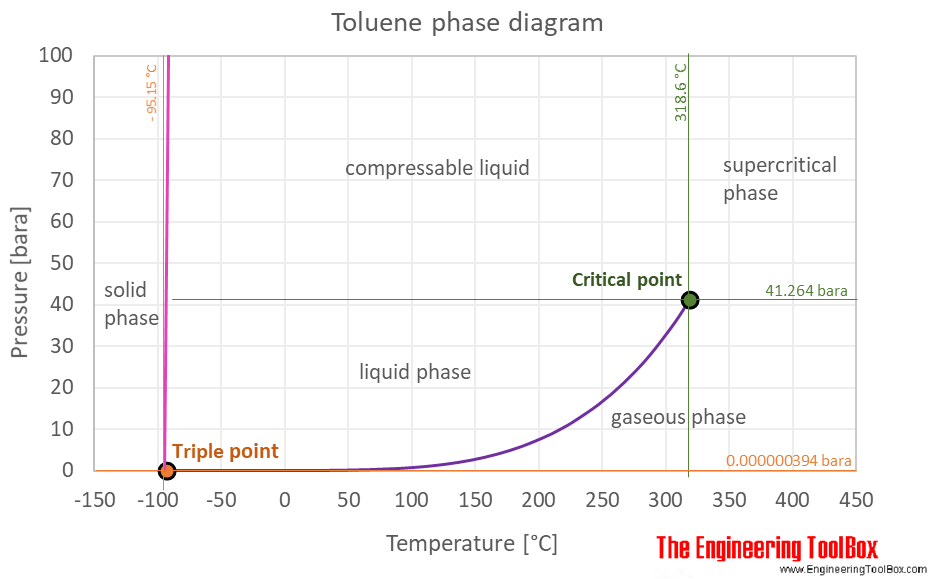
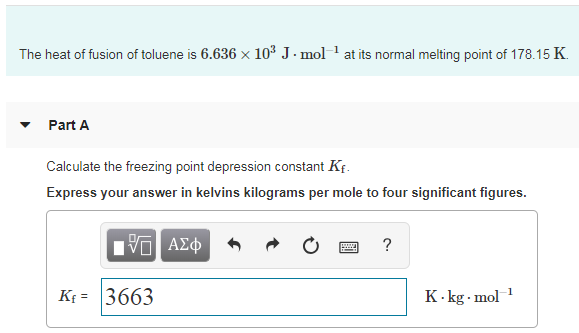
SOLVED: What is the freezing point . of the solution when 52.2 g of toluene (CzHs) is dissolved in 481 g of naphthalene? The freezing point constant for naphthalene is 7.00 %Clm.

When 1.5 gm phenol is disolved in 100 gm of toluene, decrease in freezing point is 0.56 K is observed. If its association is dimeric in nature, then find out percentage of

SOLVED: Question 32 (3 points) What is the freezing point of a solution prepared from 44.0 g ethylene glycol (CzH6O2) and 72.0 g H2O? Kf of water is 1.86"C/m. OA -17.6*C B) -
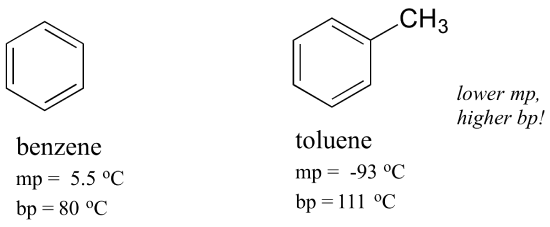
![1. The boiling point diagram for a mixture of benzene and toluene is shown below. [{Image src='fraction7304253463166023866.jpg' alt='fraction' caption=''}] a. What is the boiling point of pure toluen | Homework.Study.com 1. The boiling point diagram for a mixture of benzene and toluene is shown below. [{Image src='fraction7304253463166023866.jpg' alt='fraction' caption=''}] a. What is the boiling point of pure toluen | Homework.Study.com](https://homework.study.com/cimages/multimages/16/fraction7304253463166023866.jpg)
1. The boiling point diagram for a mixture of benzene and toluene is shown below. [{Image src='fraction7304253463166023866.jpg' alt='fraction' caption=''}] a. What is the boiling point of pure toluen | Homework.Study.com

Phenol associates in benzene to a certain extent to form a dimer. A solution containing 20 × 10^-3 kg phenol in 1 kg of benzene has its freezing point depressed by 0.69

SOLVED: What is the freezing point . of the solution when 52.2 g of toluene (CzHs) is dissolved in 481 g of naphthalene? The freezing point constant for naphthalene is 7.00 %Clm.

Freezing Point Depression - Calculate the freezing point of a solution prepared by dissolving 0 mole - Studocu
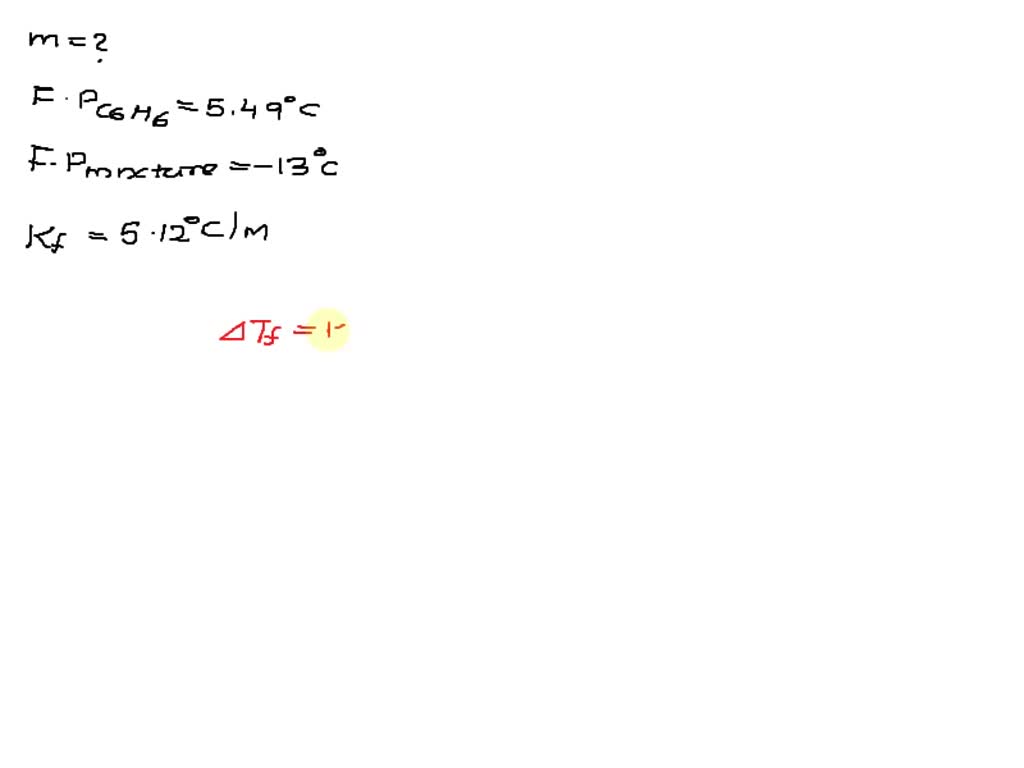
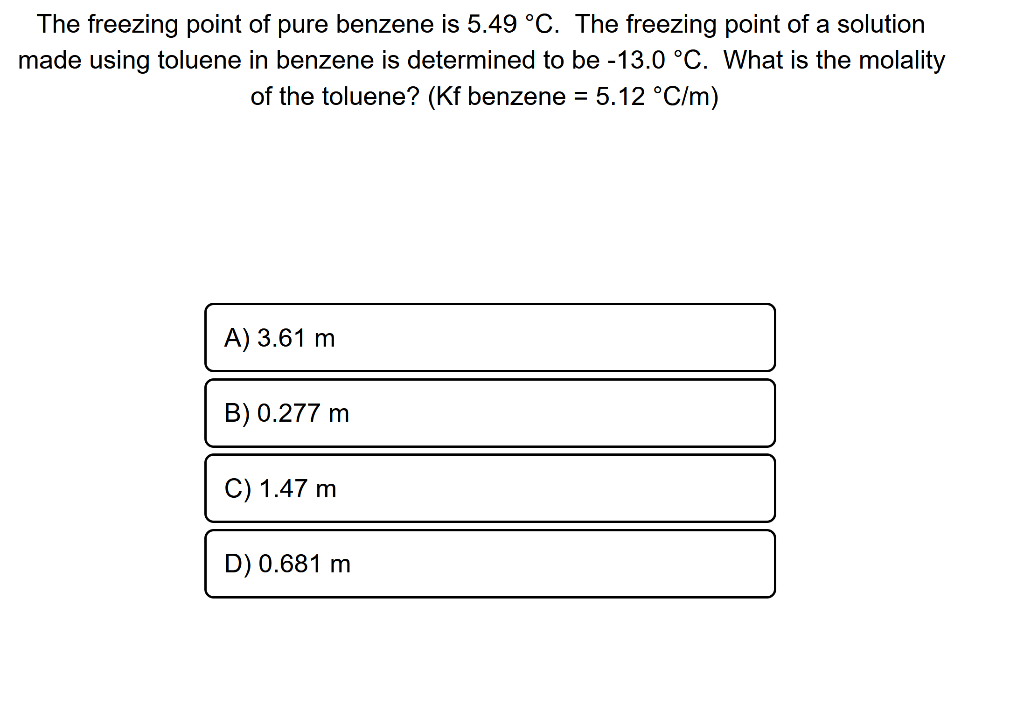
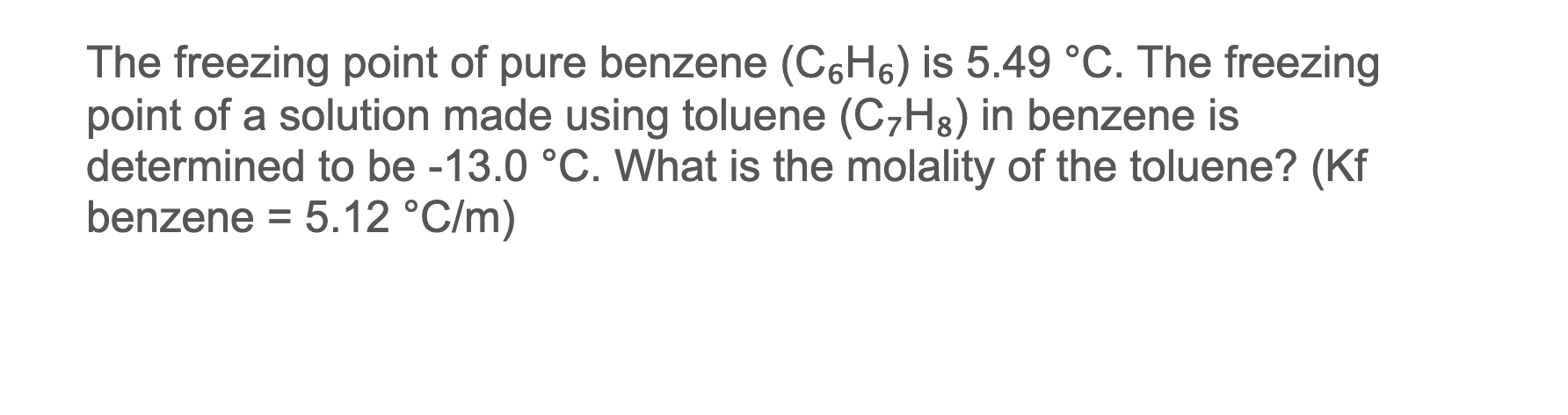
SOLVED: The freezing point of pure benzene (C₆H₆) is 5.49 °C. The freezing point of a solution made using toluene (C₇H₈) in benzene is determined to be -13.0 °C. What is the
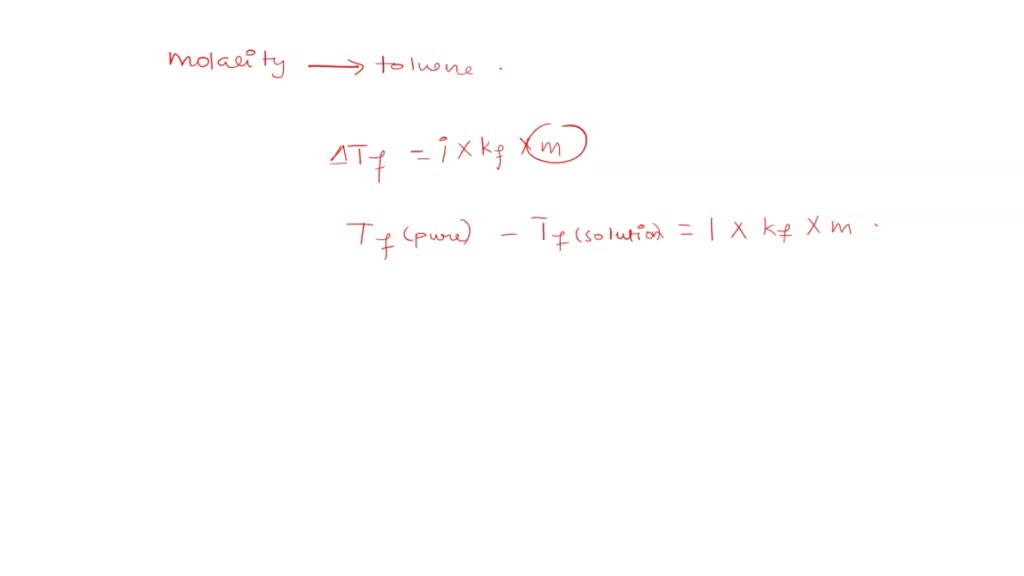
SOLVED: 1. The freezing point of pure benzene (C₆H₆) is 5.49 °C. The freezing point of a solution made using toluene (C₇H₈) in benzene is determined to be -13.0 °C. What is