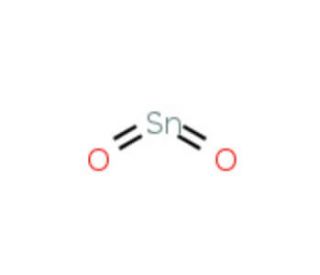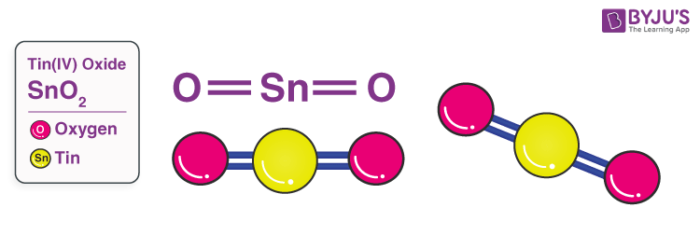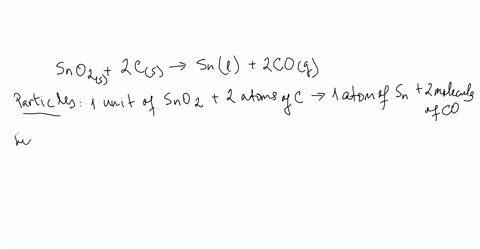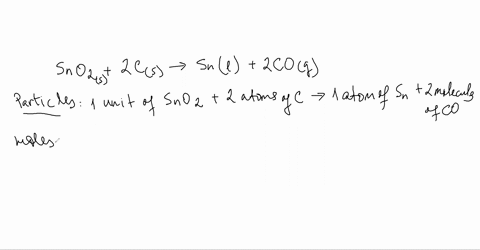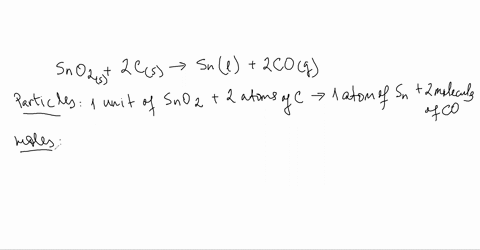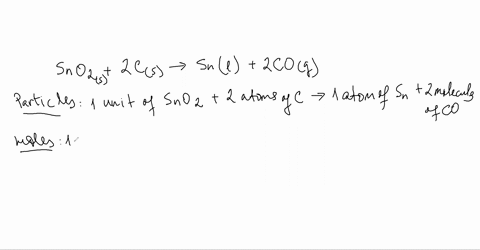
SOLVED:Smelting When tin(IV) oxide is heated with carbon in a process called smelting, the element tin can be extracted. SnO2(s)+2 C(s) →Sn(1)+2 CO(g) Interpret the chemical equation in terms of particles, moles,

CHEMISTRY NOTES Polyatomic Ions. What is the formula for…. Iron (III) chloride FeCl 3 Copper (II) nitride Cu 3 N 2 Tin (II) Oxide SnO. - ppt download

Write formulas for the following compounds: a. Mercury (II) chloride b. Nickel (II) sulphate c. ... - YouTube

Write the formula of the following compunds: (i)Tin (IV) oxide ,(ii) Copper (I) iodide ,(iii) chromium (III) oxide ,(IV) Thallium (I) suplhate ,(V) Bismuth (III) niitrate Mercury (II) chloride