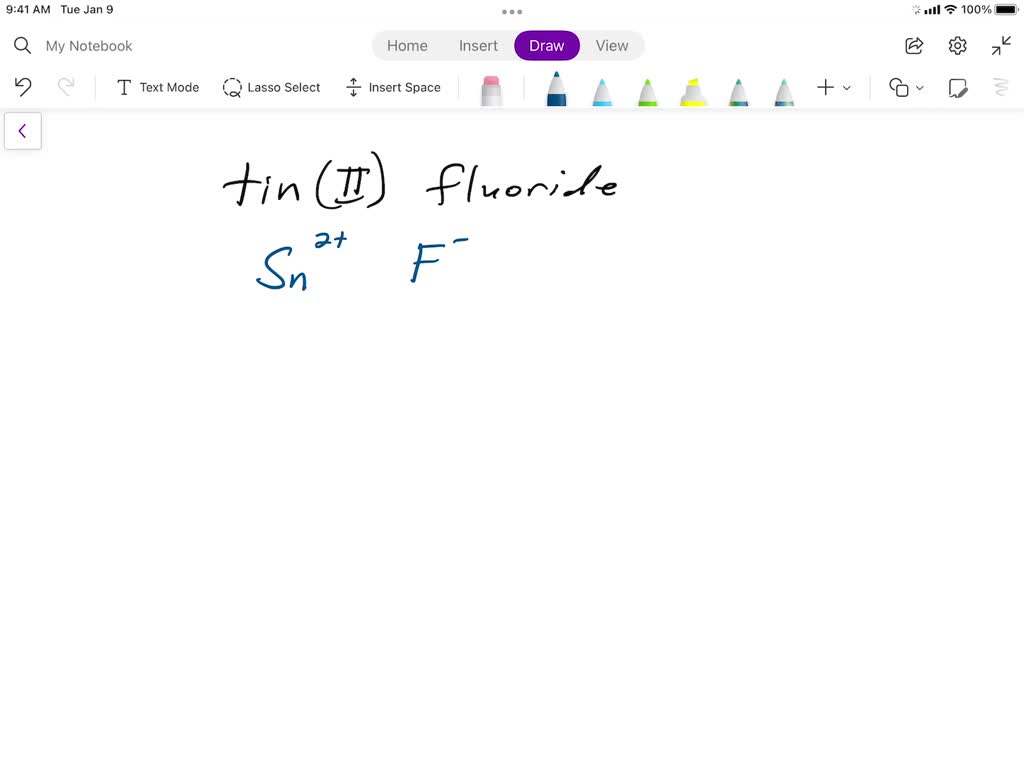
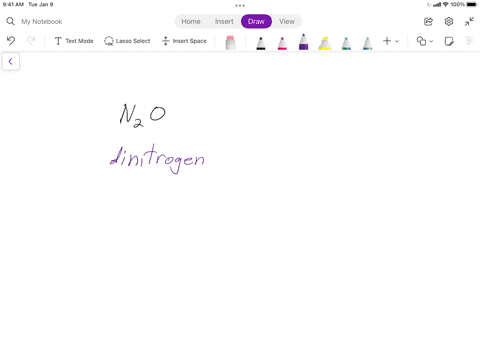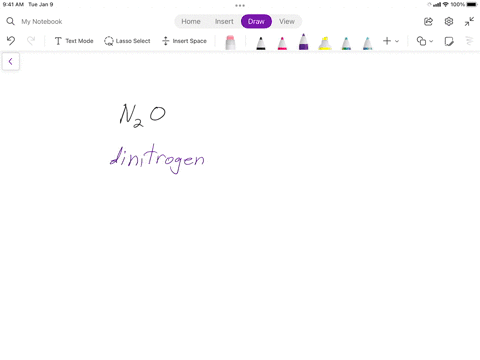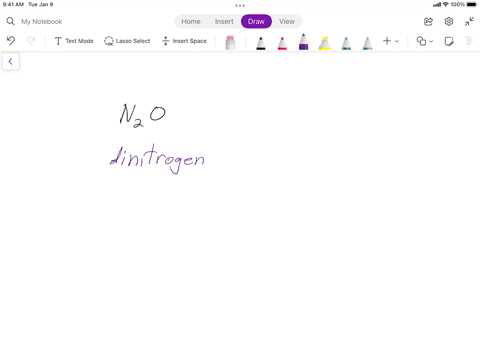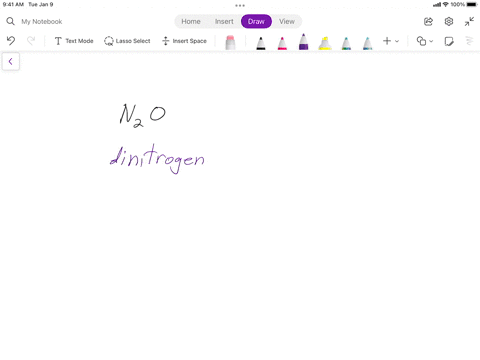
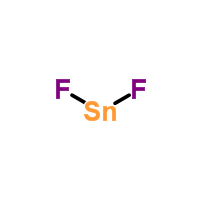
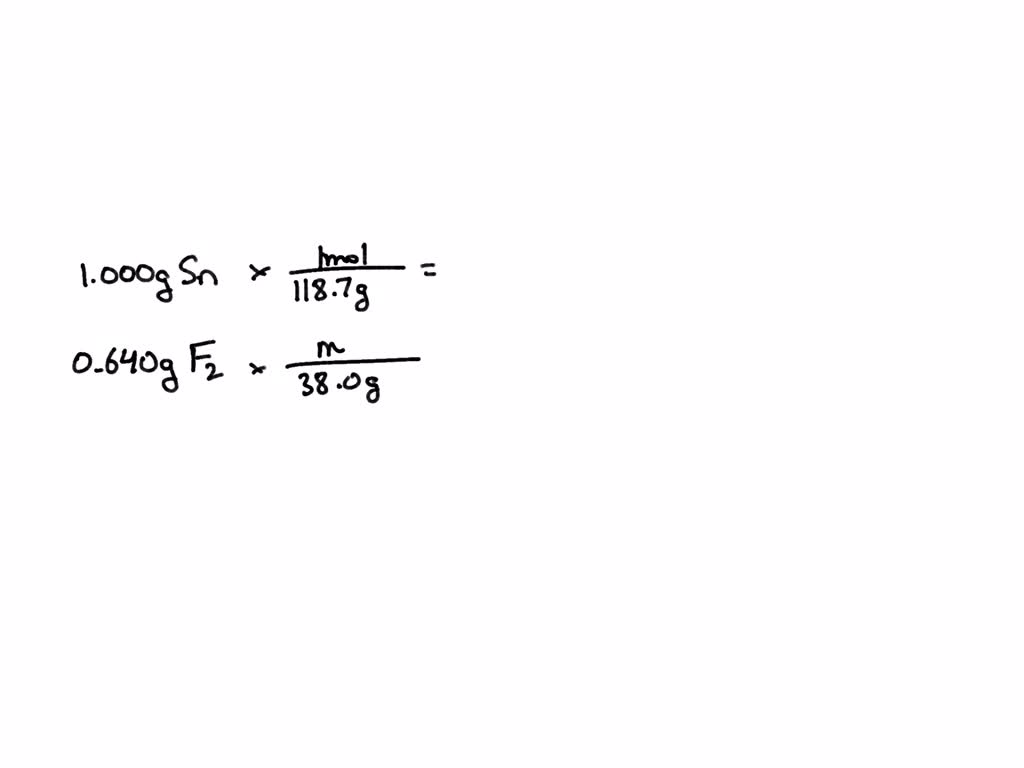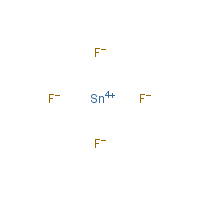SOLVED: Writing Formulas for lonic Compounds a; copper() fluoride b iron(III) chloride C mercury(l) sulfide d. nickel(ll) oxide e. iron(Il) bromide f. tin(IV) nitride g. manganese(II) iodide h lead(IV) sulfide gold(III) phosphide

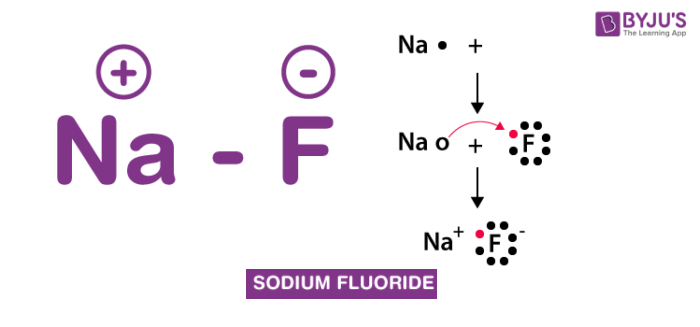
Chemistry Nomenclature Binary Ionic Compounds. What are they made of? Binary Ionic Example sodium, Na + metal chlorine, Cl - non- metal metal. - ppt download