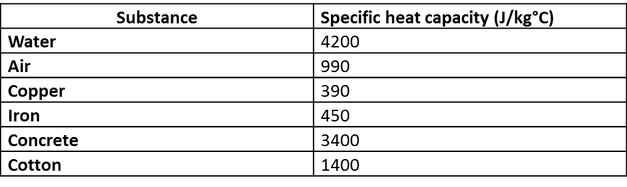
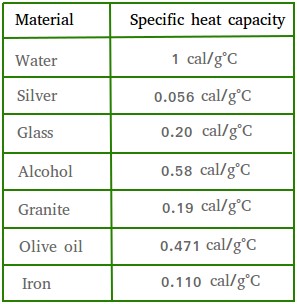
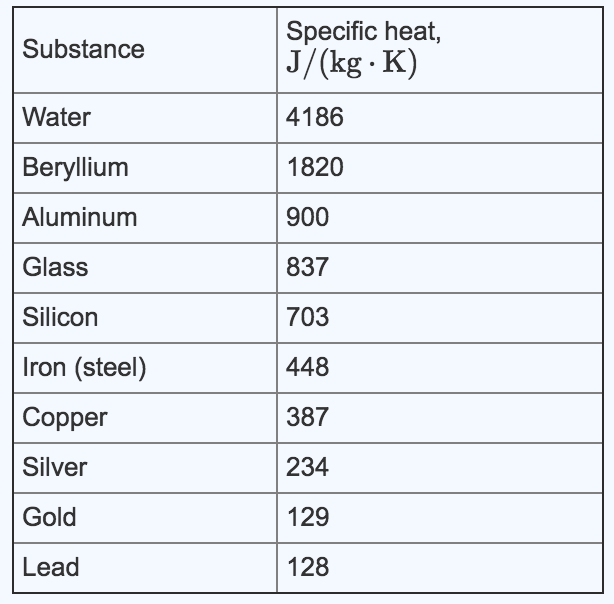
Comparison between the calculated and experimental heat capacity of... | Download Scientific Diagram

Derived mean values of the specific heat of pure iron in comparison... | Download Scientific Diagram
![PDF] Electronic Specific Heat of Iron Pnictides Based on Electron-Cooper Pair Interaction | Semantic Scholar PDF] Electronic Specific Heat of Iron Pnictides Based on Electron-Cooper Pair Interaction | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/36a34a8a61709b713d9e6e7e12f0b93504140c9c/7-Figure2-1.png)
PDF] Electronic Specific Heat of Iron Pnictides Based on Electron-Cooper Pair Interaction | Semantic Scholar

Calculate the amount of heat required to raise the temperature of 5 g of iron from `25^(@)C \"to\" - YouTube
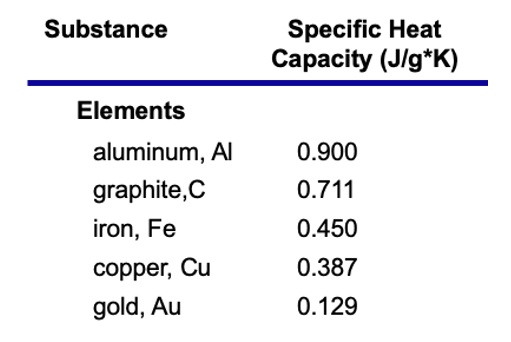
SOLVED: Substance Specific Heat Capacity (Jlg*K) Elements aluminum, Al graphite,C iron, Fe copper; Cu gold, Au 0.900 0.711 0.450 0.387 0.129
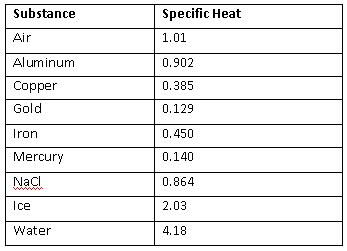
A 36.07 g sample of a substance is initially at 27.8°C. After absorbing 2639 J of heat, the temperature of the substance is 109.0°C. What is the specific heat of the substance? | Socratic
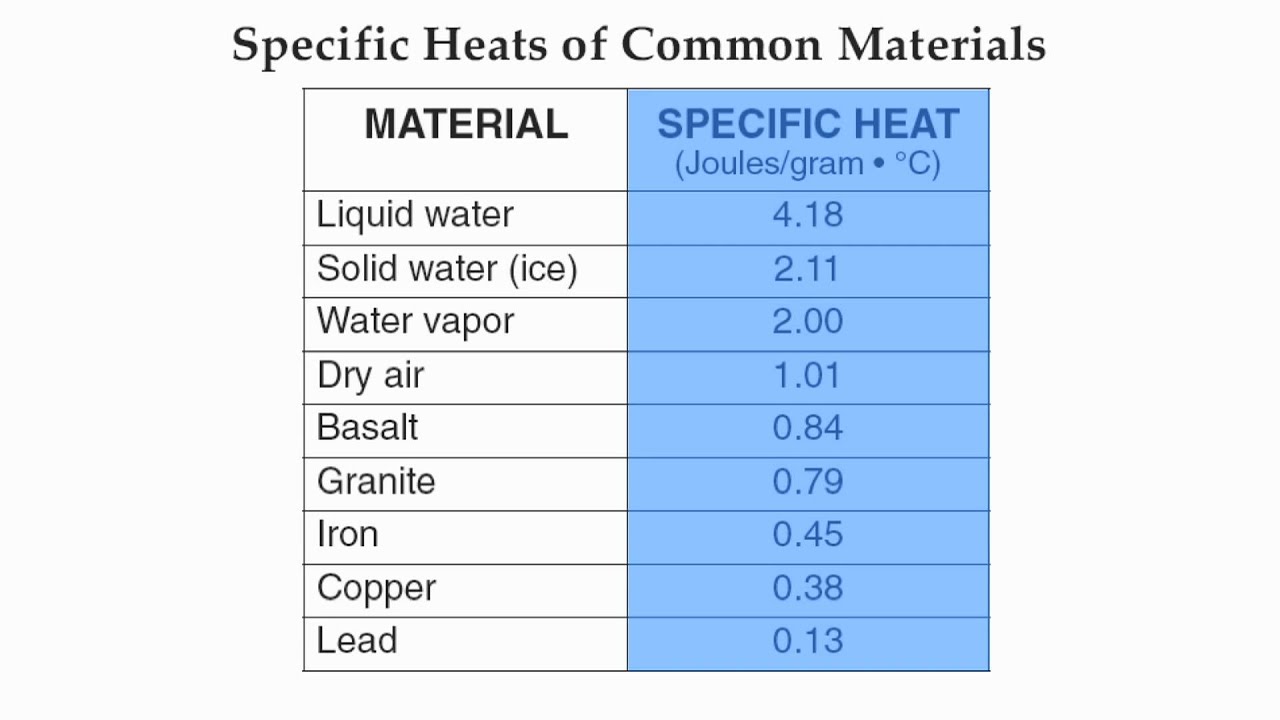
Reference Table Page 1-Specific Heat of Common Materials-Hommocks Earth Science Department - YouTube
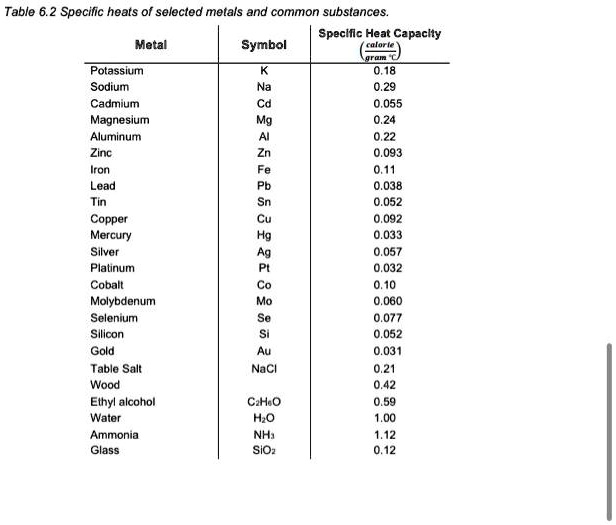
SOLVED: Table 6.2 Specific heats of selected metals and common substances: Speclllc Heat Capacly Metal Symbol Potassium Sodium Cadmium Magnesium Aluminum Zinc Iron Lead 0,18 055 093 0,11 0.038 052 092 033

Color online) Temperature-dependent specific heat capacities of (a)... | Download Scientific Diagram

Variations of specific heat (plot a) and thermal conductivity (plot b)... | Download Scientific Diagram
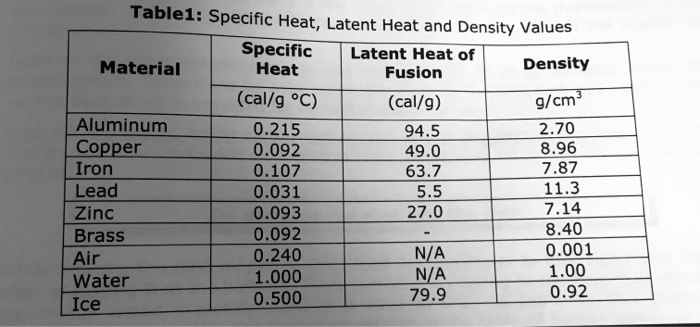
SOLVED: Table1: Specific Heat, Latent Heat ad Density Values Specific Latent Heat of Material Heat Density Fusion (cal/g 'C) (cal/g) g/cm Aluminum 0.215 94.5 2.70 Copper 0.092 49 8.96 Iron 0.107 63
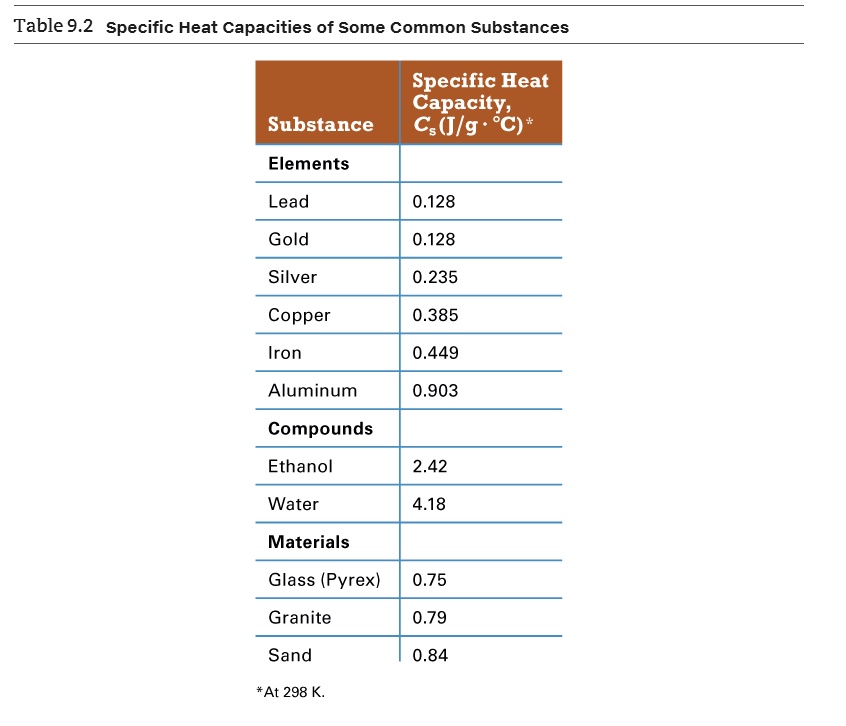
SOLVED: Table 9.2 Specific Heat Capacities of Some Common Substances Specific Heat Capacity; Cs(Jlg. *C) Substance Elements Lead 0.128 Gold 0.128 Silver 0.235 Copper 0.385 Iron 0.449 Aluminum 0.903 Compounds Ethanol 2.42

Calculate the energy required to heat 790.0g of iron from −2.6°C to 14.9°C. Assume the specific heat - Brainly.com