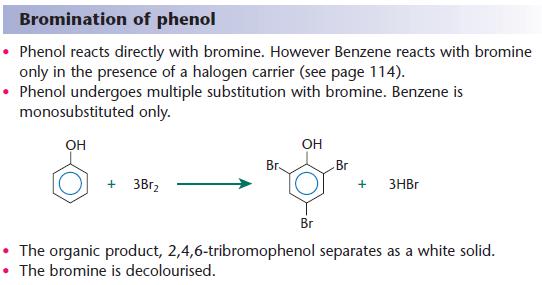
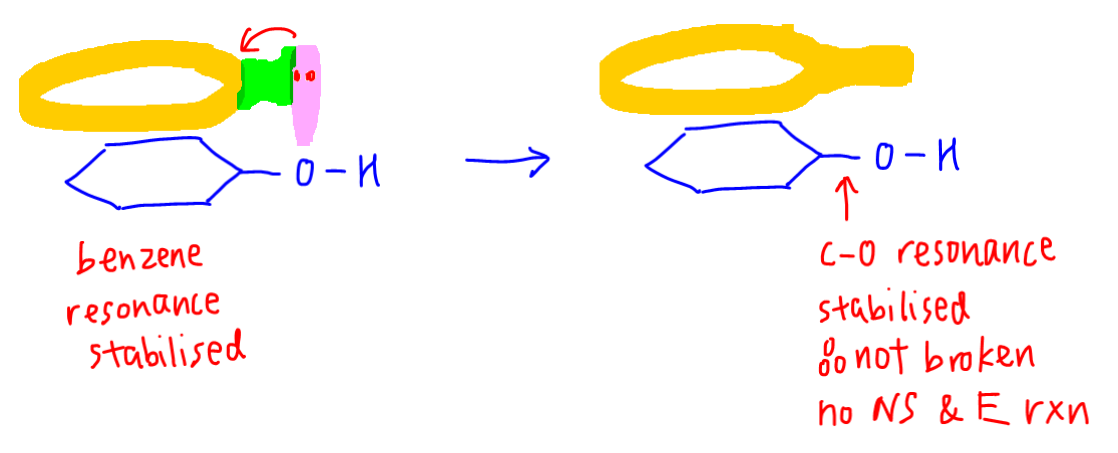
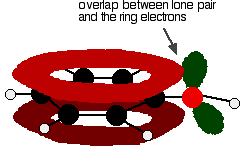
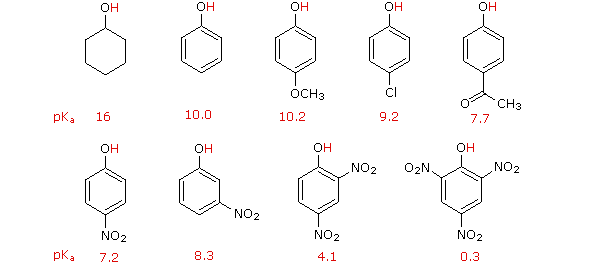
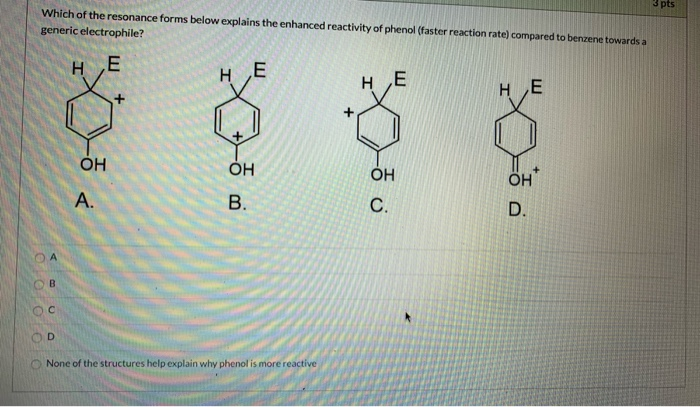
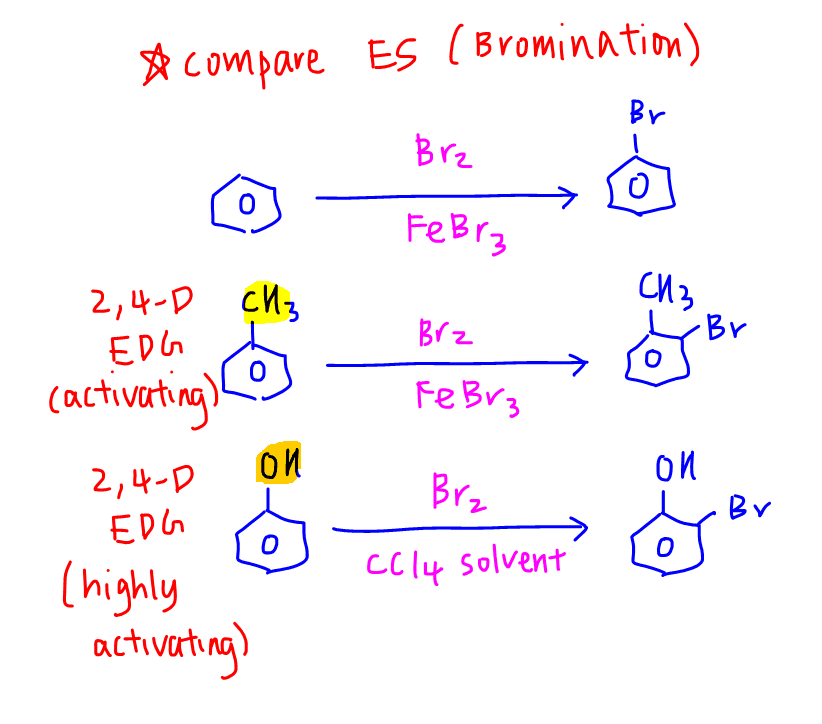
Phenol hydrogen bonding physical chemical properties electrophilic substitution chlorine bromine nitric acid acidity of phenols uses chlorophenols phenyl phenolic esters advanced A level organic chemistry revision notes doc brown
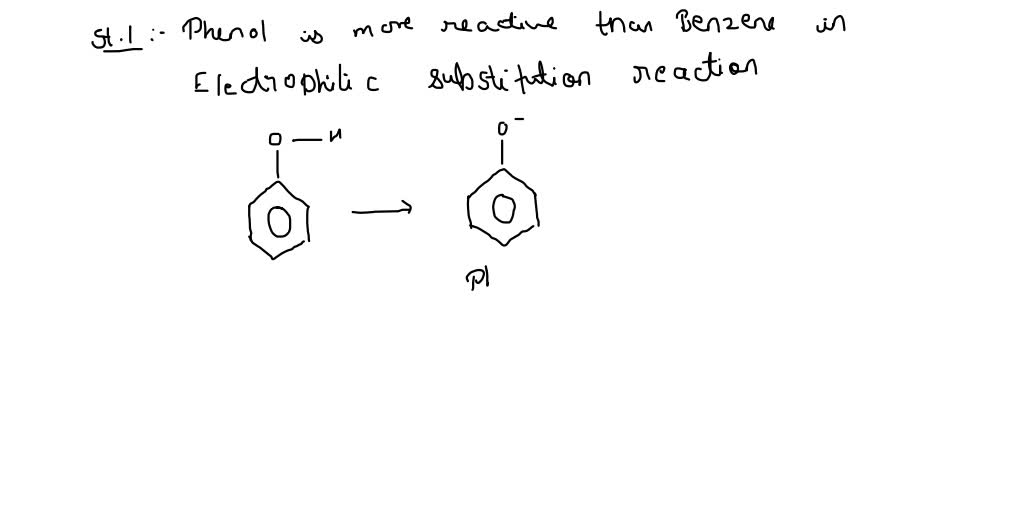
SOLVED: Statement 1 Phenol is more reactive than benzene in electrophilic substitution reactions. and Statement 2 The higher reactivity of phenol is due to the higher acidity of phenol.

Acid-promoted direct electrophilic trifluoromethylthiolation of phenols - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB02633K
Which will give electrophilic substitution reaction faster: phenol or aniline in acidic medium? - Quora
Explain why is phenoxide ion more stable than phenol towards electrophilic substitution reaction? - Quora

Phenol hydrogen bonding physical chemical properties electrophilic substitution chlorine bromine nitric acid acidity of phenols uses chlorophenols phenyl phenolic esters advanced A level organic chemistry revision notes doc brown
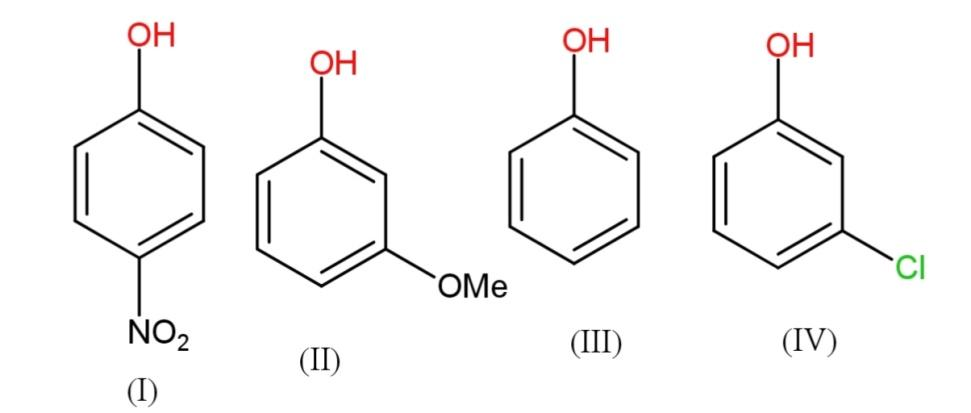
The correct increasing order of reactivity for the following molecules towards electrophilic aromatic substitution is:\n \n \n \n \n A.$I IV II III$ B. $I IV III II$C. $I III II IV$D. $