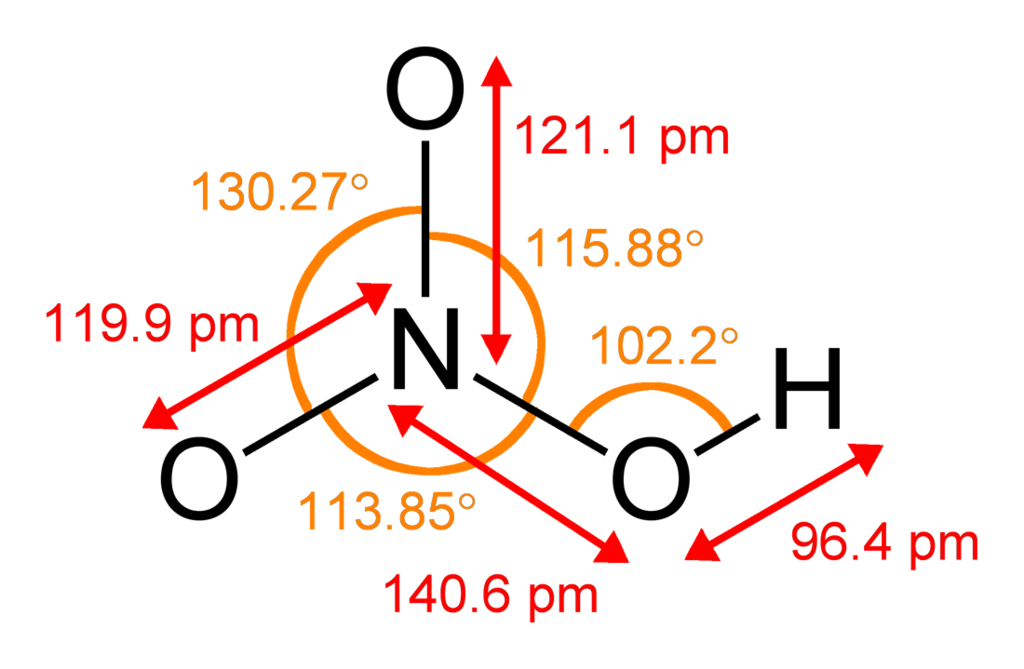
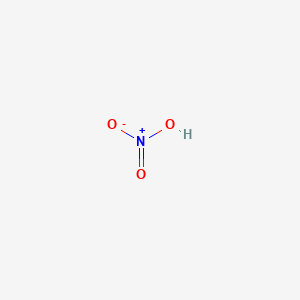
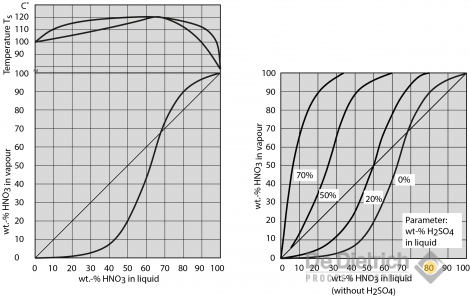
Homogeneous freezing of single sulfuric and nitric acid solution drops levitated in an acoustic trap - ScienceDirect
Effects of nitrogen tetroxide and water concentration on freezing point and ignition delay of fuming nitric acid - UNT Digital Library

Homogeneous Freezing of Concentrated Aqueous Nitric Acid Solutions at Polar Stratospheric Temperatures | The Journal of Physical Chemistry A

228. The heat of nitrolysis of hexamine in nitric acid - Journal of the Chemical Society (Resumed) (RSC Publishing)

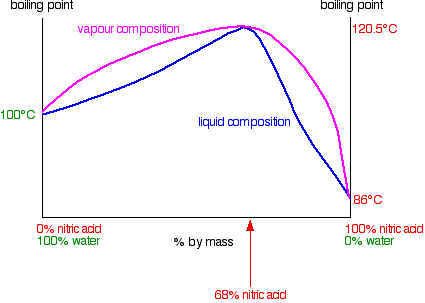
Phase Diagram of the Nitric Acid/Water System: Implications for Polar Stratospheric Clouds | The Journal of Physical Chemistry A
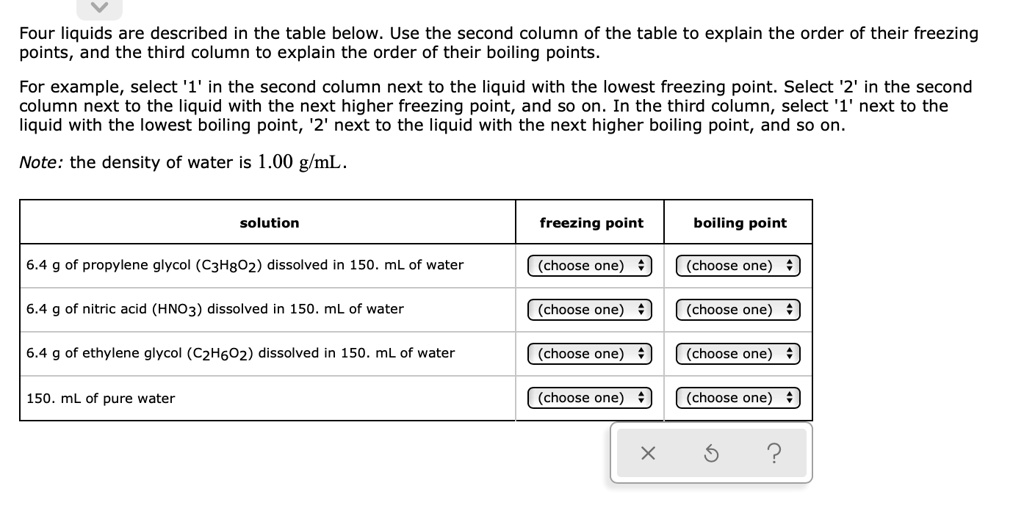
SOLVED: Four liquids are described in the table below: Use the second column of the table to explain the order of their freezing points, and the third column to explain the order

The state diagram of aqueous citric acid solution droplets. The plot... | Download Scientific Diagram

Homogeneous Freezing of Concentrated Aqueous Nitric Acid Solutions at Polar Stratospheric Temperatures | The Journal of Physical Chemistry A

SOLVED: The experimentally determined freezing point of 1.00% solution (mass %) of nitric acid (HNOs) is 0.56 'C Calculate the molality of the solution;, determine the calculated freezing point; and then calculate

a Corrosion of Ti and Ti alloys in boiling nitric acid, for a total... | Download Scientific Diagram