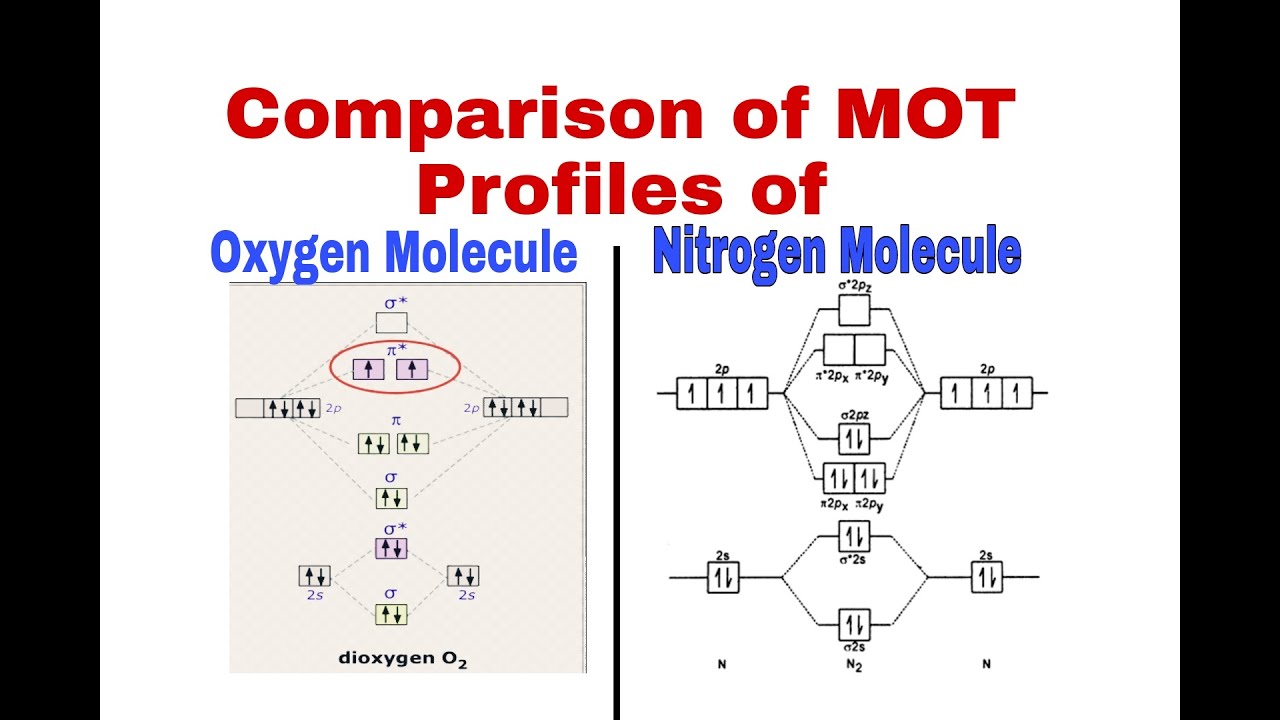
Comparison between MOT Profiles of Nitrogen & Oxygen| N2, Be, B & O2,F2|Energy of sigma 2px |F.sc 1 - YouTube

write electronic configuration of N2 and O2+ MOT - Chemistry - Chemical Bonding and Molecular Structure - 11356343 | Meritnation.com
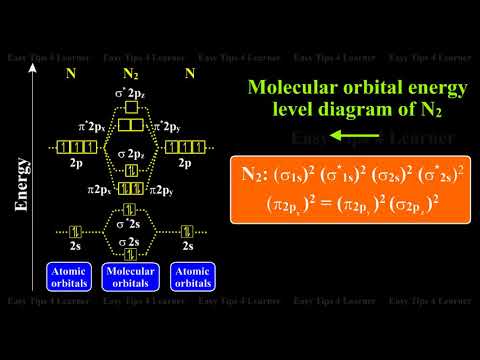
Nitrogen Molecule : Molecular Orbital Energy Level Diagrams | Diatomic Homonuclear Molecules - YouTube
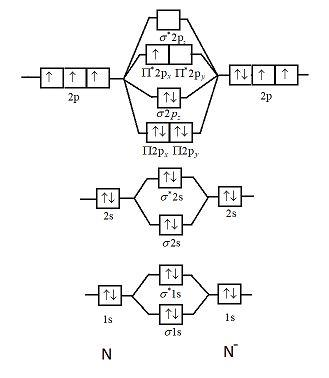
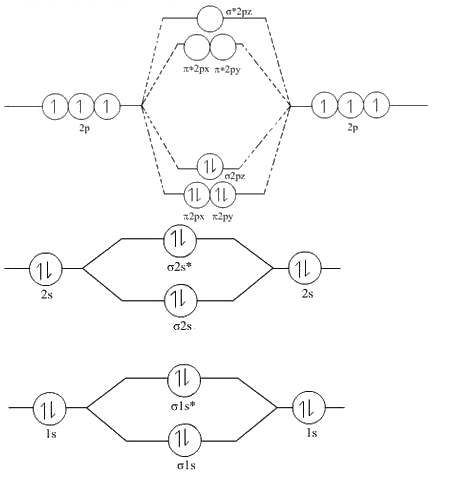
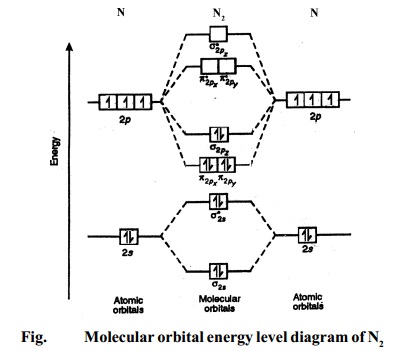
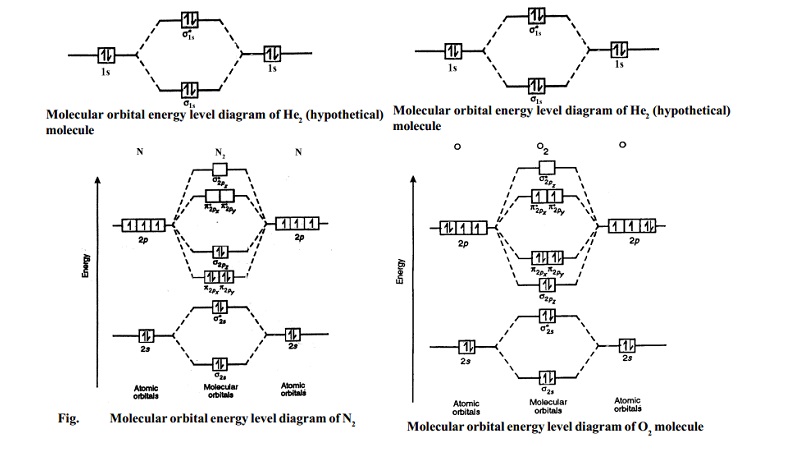
Draw the molecular orbital diagram of ${N_2},N_2^ + ,N_2^ - $. Write their electronic configuration, find the bond order and predict their magnetic behavior. Arrange the above in increasing order of bond length
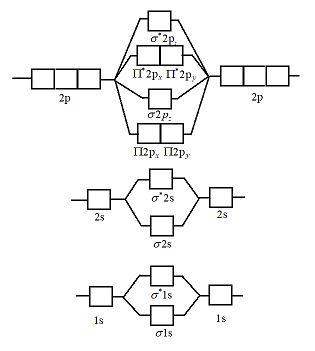
Draw the molecular orbital diagram of ${N_2},N_2^ + ,N_2^ - $. Write their electronic configuration, find the bond order and predict their magnetic behavior. Arrange the above in increasing order of bond length
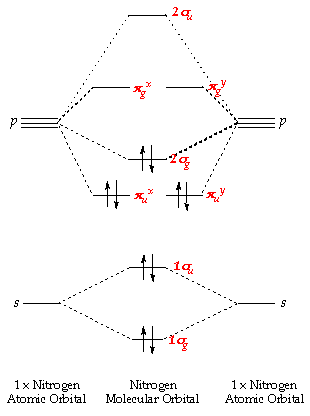
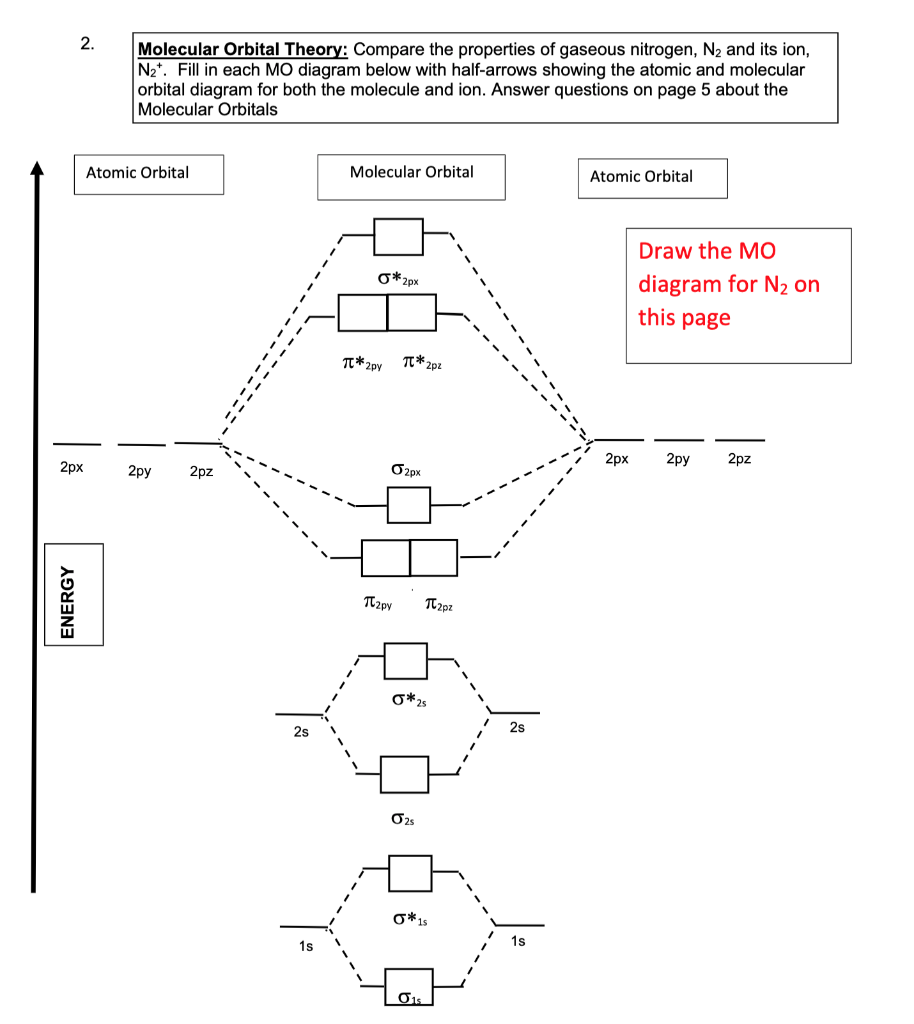
What are the molecular orbital configurations for N_2^+, N_2 ^(2+), N_2, N_2^-, and N_2^(2-)? | Socratic

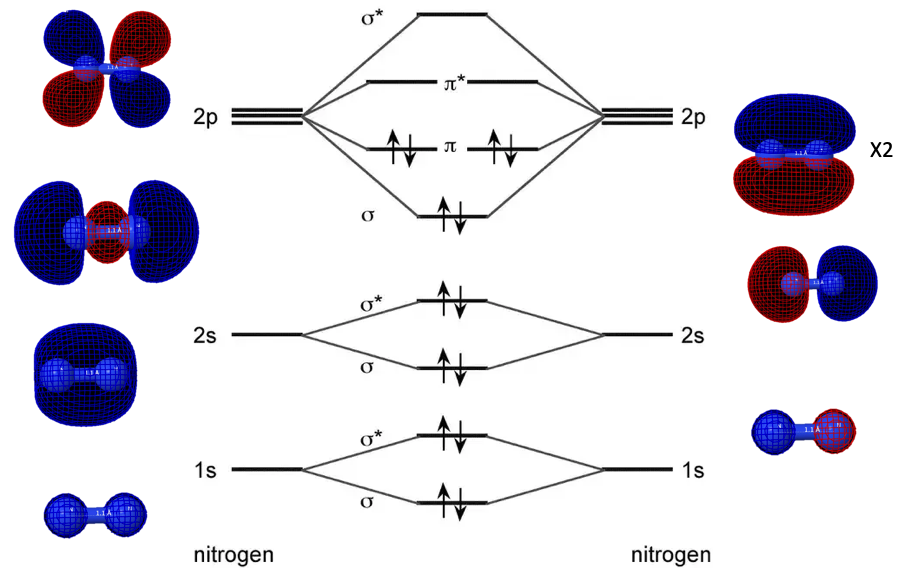
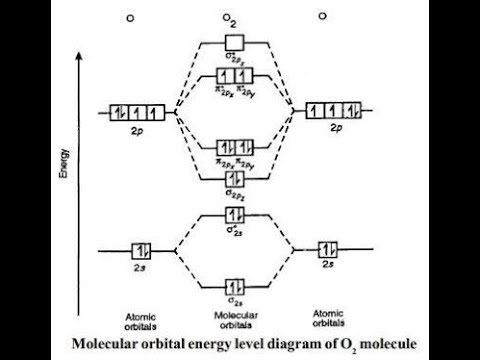
orbitals - What is the origin of the differences between the MO schemes of O₂ and N₂? - Chemistry Stack Exchange
Explain the formation of the following molecules on the basis of MOT. Also find the bond order : N2. - Sarthaks eConnect | Largest Online Education Community