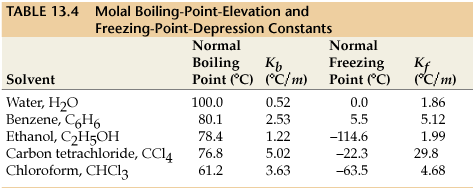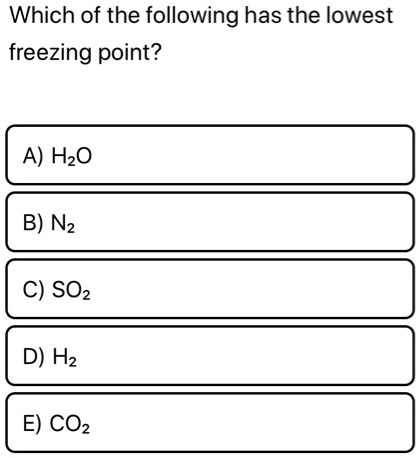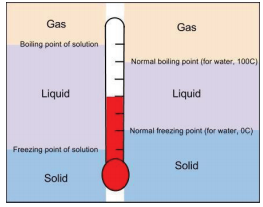
13.9: Freezing Point Depression and Boiling Point Elevation- Making Water Freeze Colder and Boil Hotter - Chemistry LibreTexts

Rank the following aqueous solutions from highest to lowest freezing point: 0.1 m FeCl3, 0.30 m glucose - Brainly.com
The solution which has lowest freezing point is Options: Should have chosen 1 M CaF2 0.01 M CaSO4 Wrong 0.005 M KI 0.1 M Al2 (SO4)3