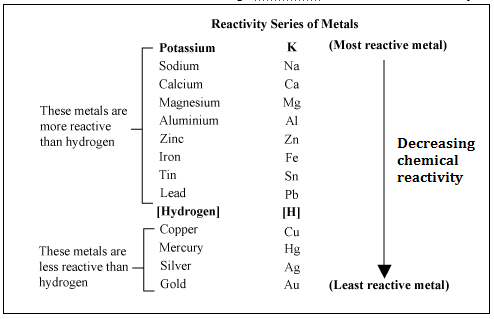
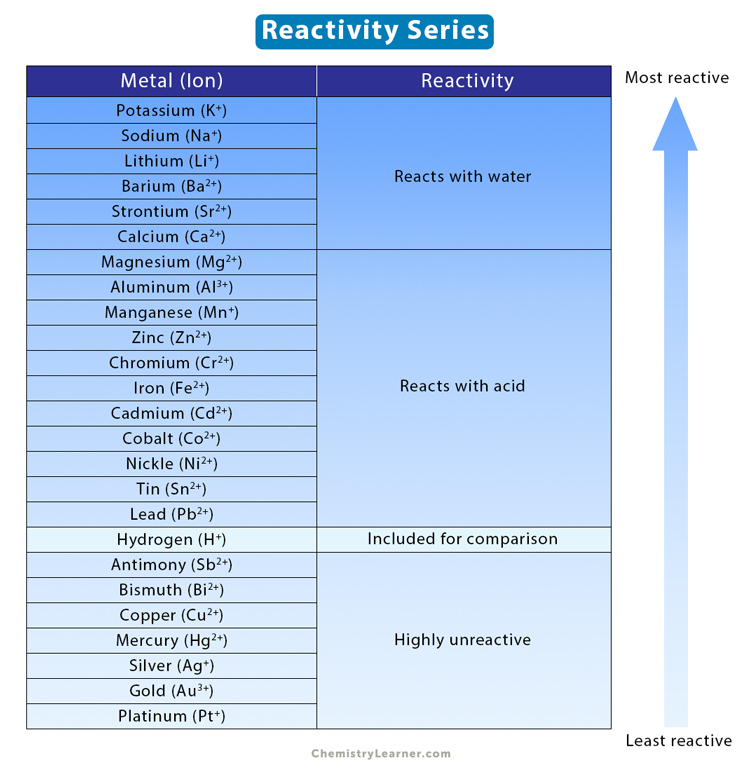
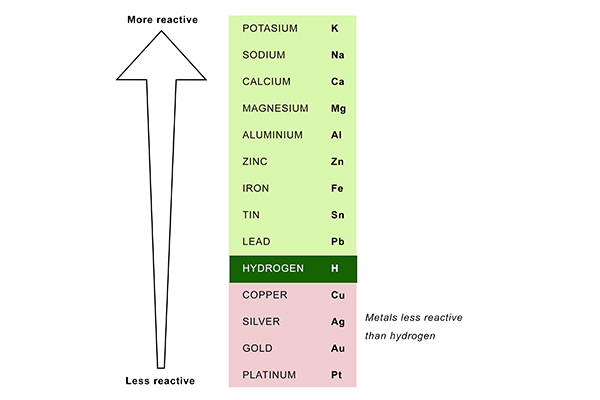
A metal wants to lose electrons, so if it is more electropositive, it is more reactive. So why is copper less reactive than hydrogen? Copper is more electropositive than hydrogen. - Quora

Arrange the given metals in the decreasing order of their reactivity.Aluminium, sodium, gold, copper, and iron.


![Reactivity Series of Metals - Chart [and How to remember] - Teachoo Reactivity Series of Metals - Chart [and How to remember] - Teachoo](https://d1avenlh0i1xmr.cloudfront.net/b0a28f61-1fb3-456e-8ae8-e110e8999f99/reactivity-series-01.jpg)