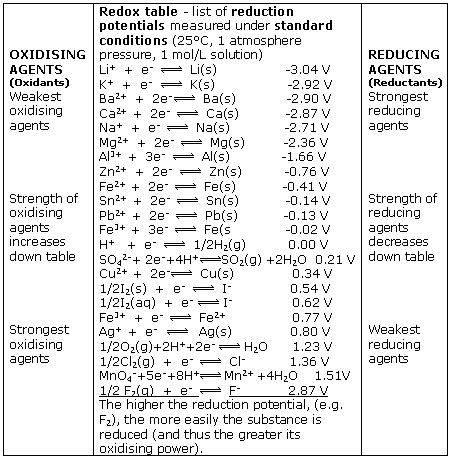
Chemistry - ELECTROCHEMICAL SERIES AND ITS APPLICATION:- A list of elements arranged in order on the basis of their standard reduction potential or oxidation potential is called electrochemical series. EXPLAINATION:- Different elements
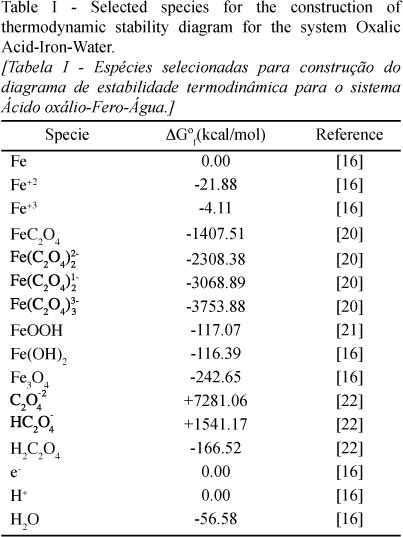
SciELO - Brasil - Thermodynamic analysis of stability in iron removal from kaolin by using oxalic acid Thermodynamic analysis of stability in iron removal from kaolin by using oxalic acid

Standard reduction potential of aluminum, calcium, iron, indium and... | Download Scientific Diagram
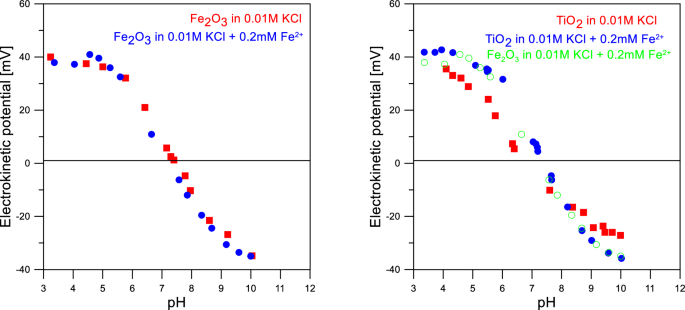
Fe2+ adsorption on iron oxide: the importance of the redox potential of the adsorption system | SpringerLink

Figure 4 from Thermodynamic Characterization of Iron Oxide-Aqueous Fe(2+) Redox Couples. | Semantic Scholar

Redox equilibria of iron oxides in aqueous-based magnetite dispersions: Effect of pH and redox potential - ScienceDirect
![PDF] Atomic resolution structures of rieske iron-sulfur protein: role of hydrogen bonds in tuning the redox potential of iron-sulfur clusters. | Semantic Scholar PDF] Atomic resolution structures of rieske iron-sulfur protein: role of hydrogen bonds in tuning the redox potential of iron-sulfur clusters. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/662c7bf260df151b4ef3677ea9876889b3502b01/3-Table1-1.png)
PDF] Atomic resolution structures of rieske iron-sulfur protein: role of hydrogen bonds in tuning the redox potential of iron-sulfur clusters. | Semantic Scholar

Determining Redox Potentials of the Iron–Sulfur Clusters of the AdoMet Radical Enzyme Superfamily - ScienceDirect

Complexation Effect on Redox Potential of Iron(III)−Iron(II) Couple: A Simple Potentiometric Experiment | Journal of Chemical Education

Range of redox potentials found amongst the different types of Fe À S... | Download Scientific Diagram

Table 1 from Redox potentials of oxoiron(IV) porphyrin π-cation radical complexes: participation of electron transfer process in oxygenation reactions. | Semantic Scholar

Benchmark Study of Redox Potential Calculations for Iron–Sulfur Clusters in Proteins | Inorganic Chemistry