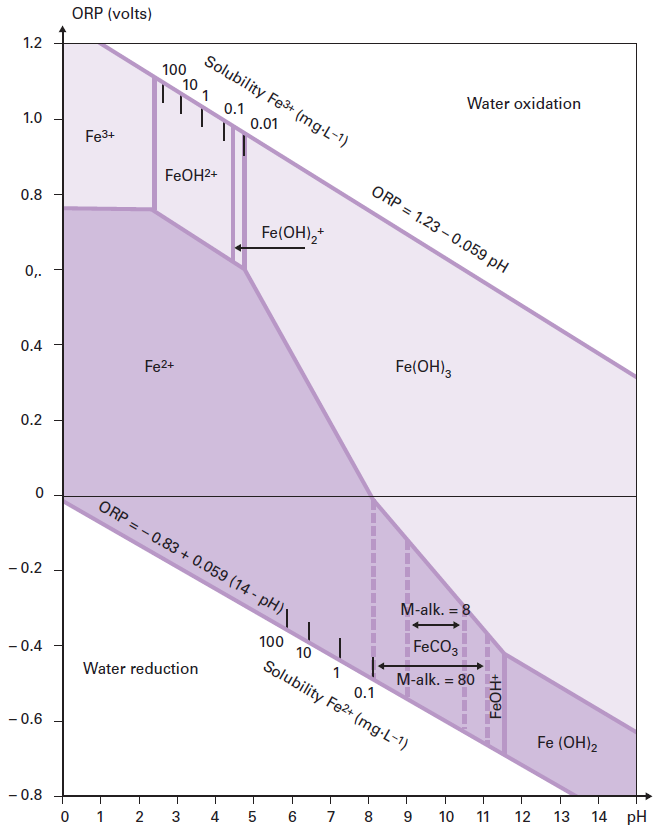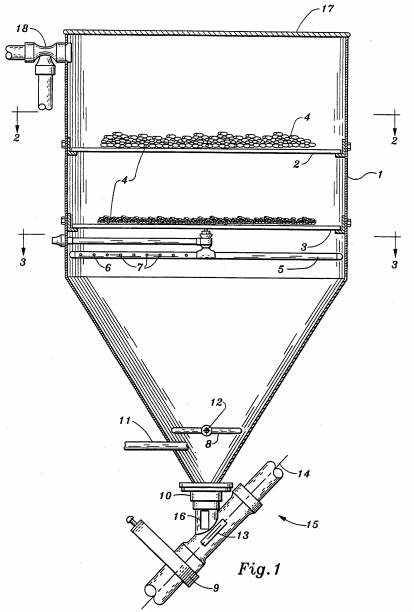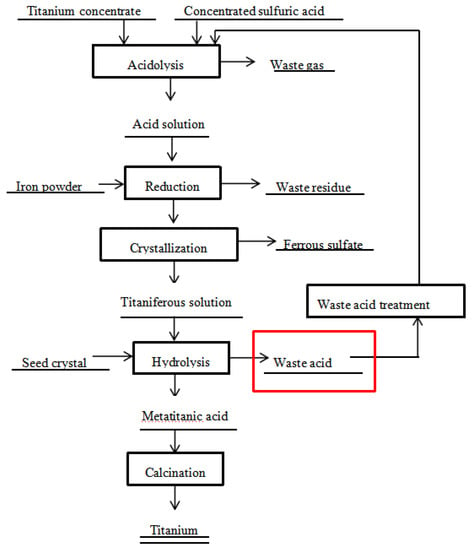
Metals | Free Full-Text | Preparation of Doped Iron Phosphate by Selective Precipitation of Iron from Titanium Dioxide Waste Acid

Precipitation reaction. Iron (III) hydroxide (brown) being precipitated from a solution by the addition of sodium hydroxide from a dropper (top). Prec Stock Photo - Alamy

Diffusion and precipitation processes in iron-based silica gardens - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C6CP02107G

Iron (II) hydroxide precipitate formed by adding sodium hydroxide (NaOH) to a solution containing iron (II) ions. Iron (II) hydroxide (Fe(OH)2) is pre Stock Photo - Alamy
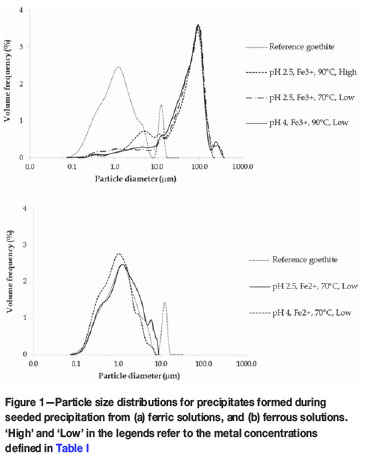
Characterization of precipitate formed during the removal of iron and precious metals from sulphate leach solutions

Chemical reaction-Rusty red iron(III) hydroxide precipitate (Fe(OH)3) in test tube formed reaction between iron(III) chloride (FeCl3) & sodium carbonate (Na2CO3): FeCl3+Na2CO3+H2O ->Fe(OH)3+NaCl+CO2 foto de Stock | Adobe Stock

Biohydrometallurgical iron oxidation and precipitation: Part I — Effect of pH on process performance - ScienceDirect

science chemical reaction precipitation iron (iii) hydroxide | Fundamental Photographs - The Art of Science

Efficient heterogeneous precipitation and separation of iron in copper-containing solution using dolomite - ScienceDirect

Sequential Precipitation of Iron, Copper, and Zinc from Wastewater for Metal Recovery | Journal of Environmental Engineering | Vol 145, No 1