zinc iron (II) iron (III) aluminum silver lead (IV) chloride ZnCl2 acetate nitrate oxide nitride sulfate chlorite phosphate
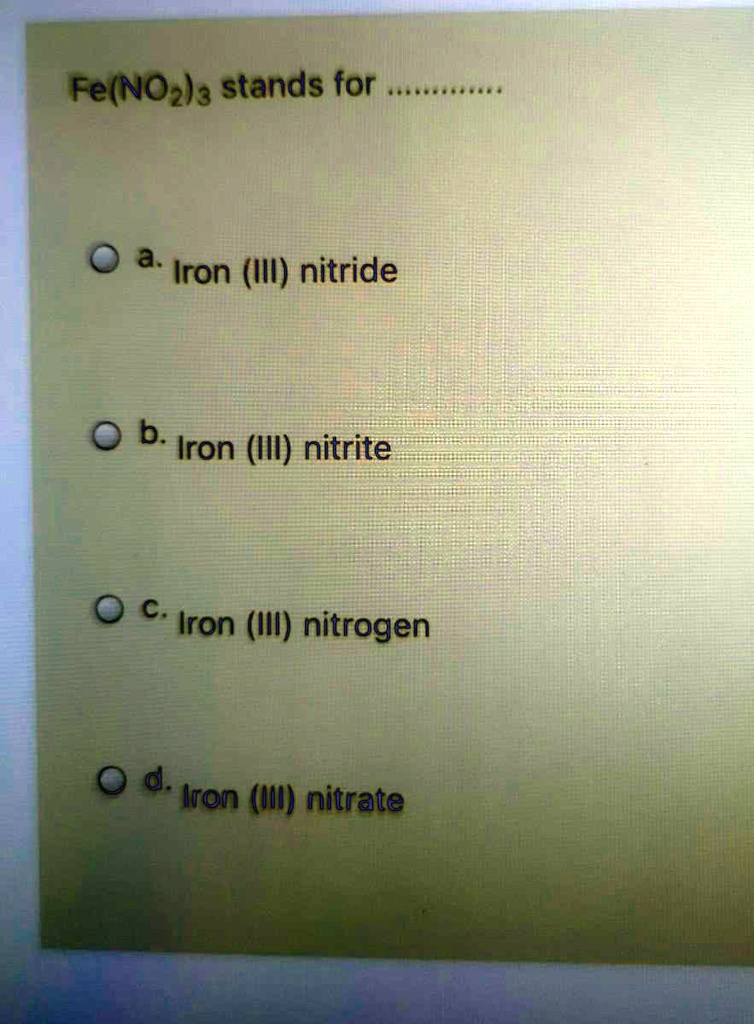
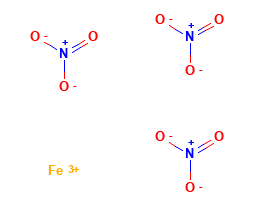
SOLVED: Fe(NOzla stands for a. Iron (III) nitride b: Iron (III) nitrite C: Iron (III) nitrogen d. Iron (III) nitrate
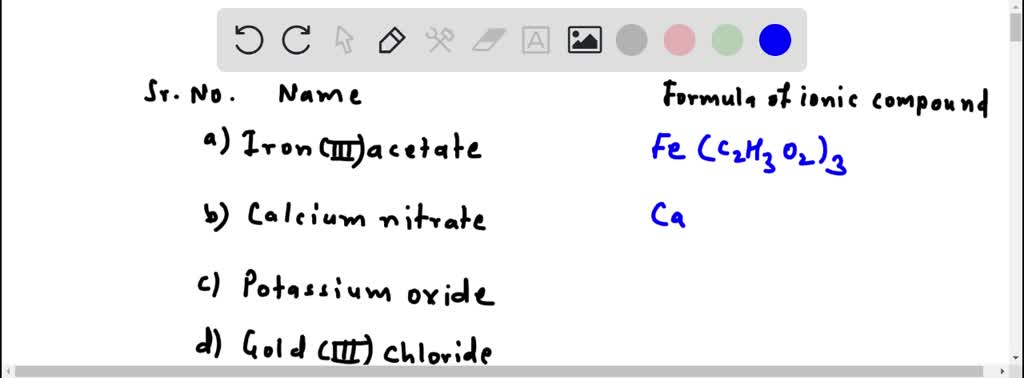
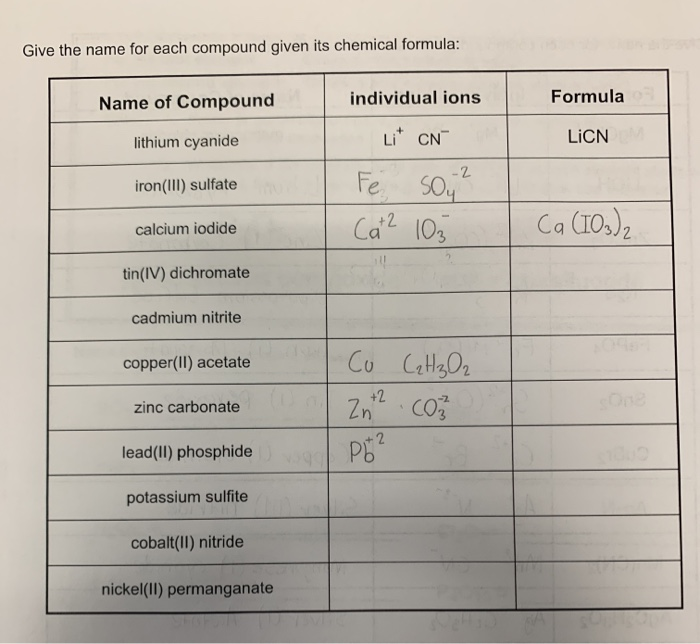
SOLVED:Write the formulas of the following ionic compounds. (a) iron(III) acetate (b) calcium nitrate (c) potassium oxide (d) gold(III) chloride (e) barium nitride

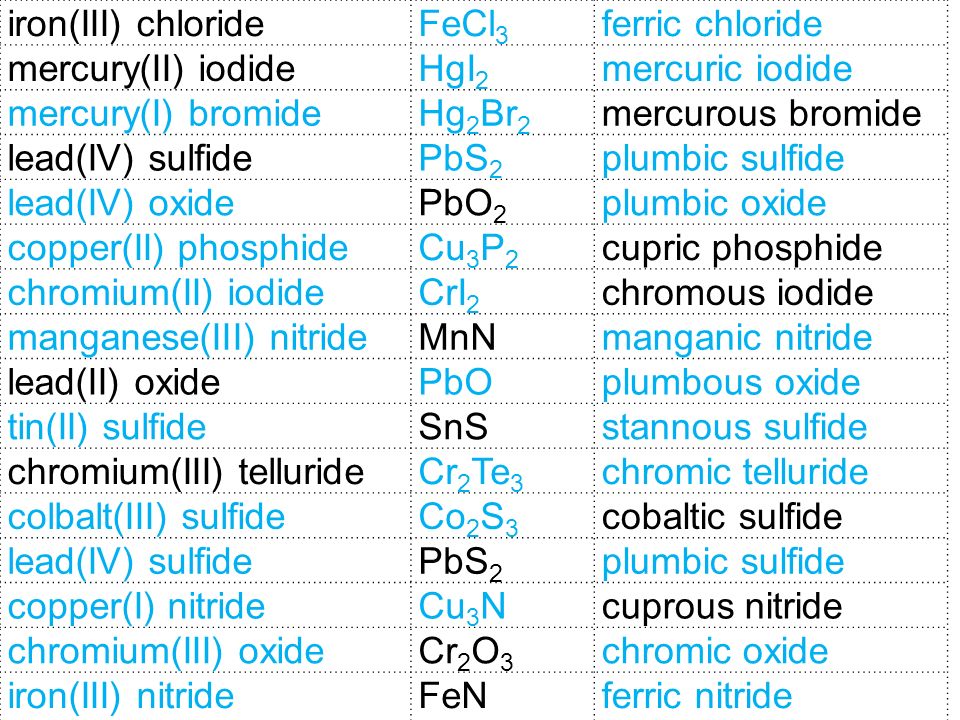
CHEMISTRY NOTES Polyatomic Ions. What is the formula for…. Iron (III) chloride FeCl 3 Copper (II) nitride Cu 3 N 2 Tin (II) Oxide SnO. - ppt download

SOLVED: Write the formula for each of the following ionic compounds: a. zinc bromide b. iron(III) sulfide c. manganese(IV) oxide d. chromium(III) iodide e. lithium nitride f. gold(I) oxide

Entry Task: Block 1 Nov. 13 th / 14 th NEW ENTRY TASK SHEET Provide the names or formulas for the following: Lead IV sulfide Zn 3 P 2 Mercury I nitride. - ppt download
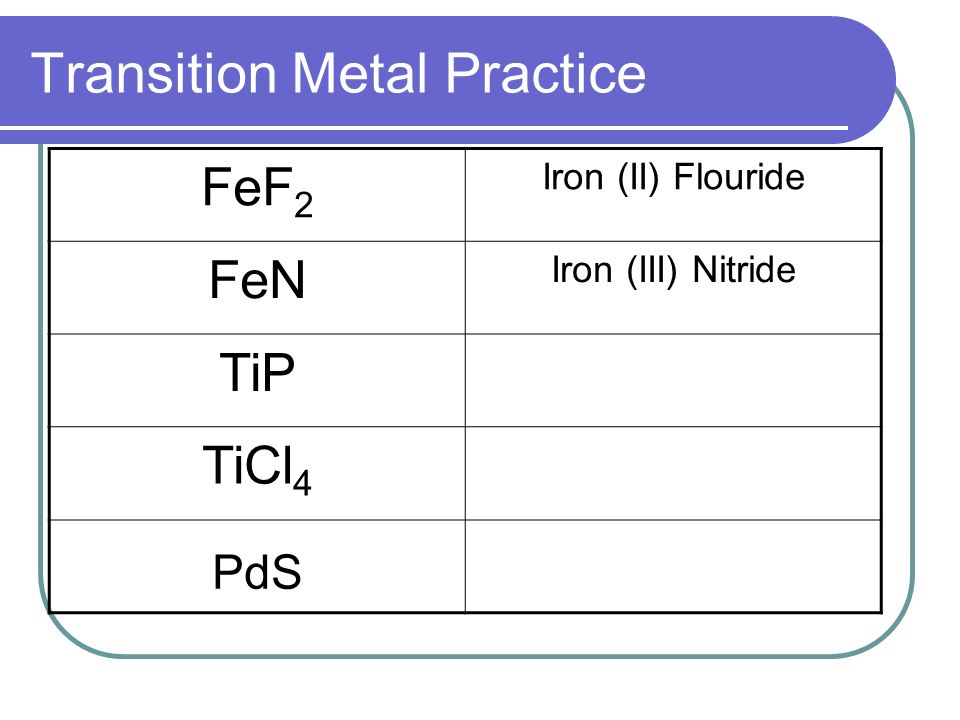
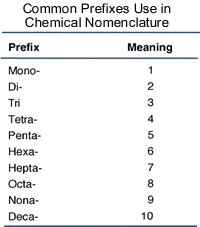
Nomenclature Ionic Compounds. Topics Ionic Nomenclature Standard Transition Metals (multivalent) Polyatomic Ions. - ppt download







![Solved The compound [iron(II)nitride] has the formula: | Chegg.com Solved The compound [iron(II)nitride] has the formula: | Chegg.com](https://media.cheggcdn.com/study/1ee/1ee06213-ac64-4042-9181-f31529cfe27d/image)