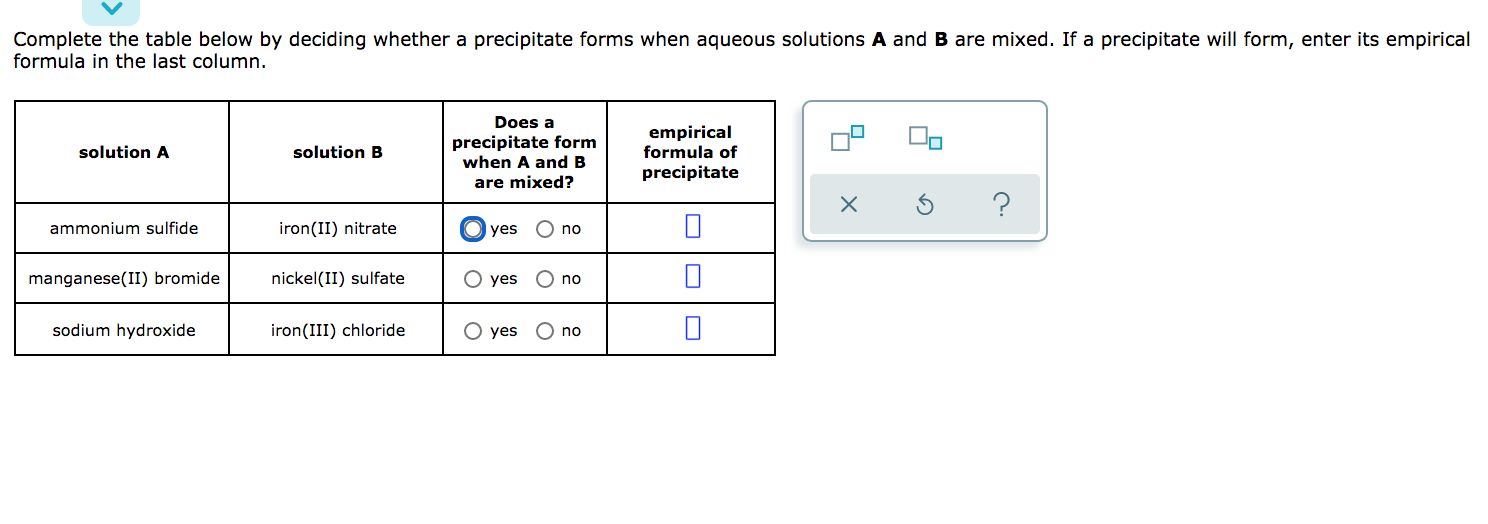
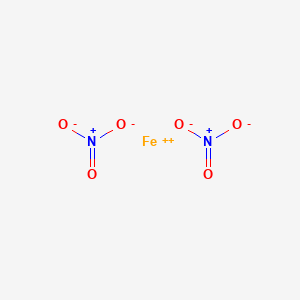
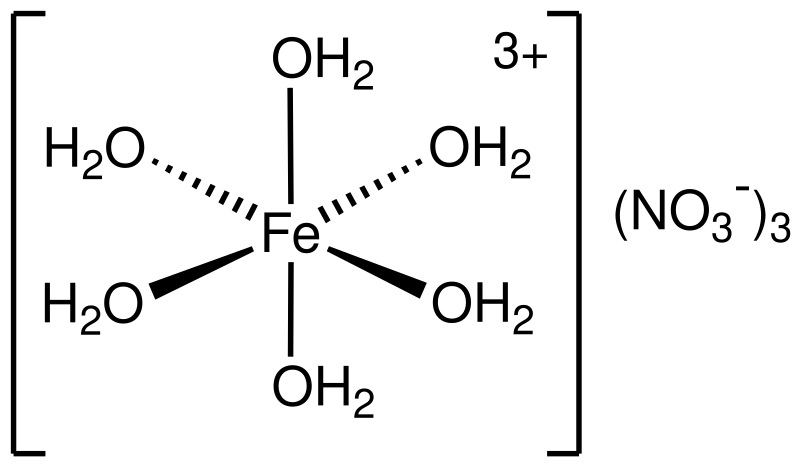
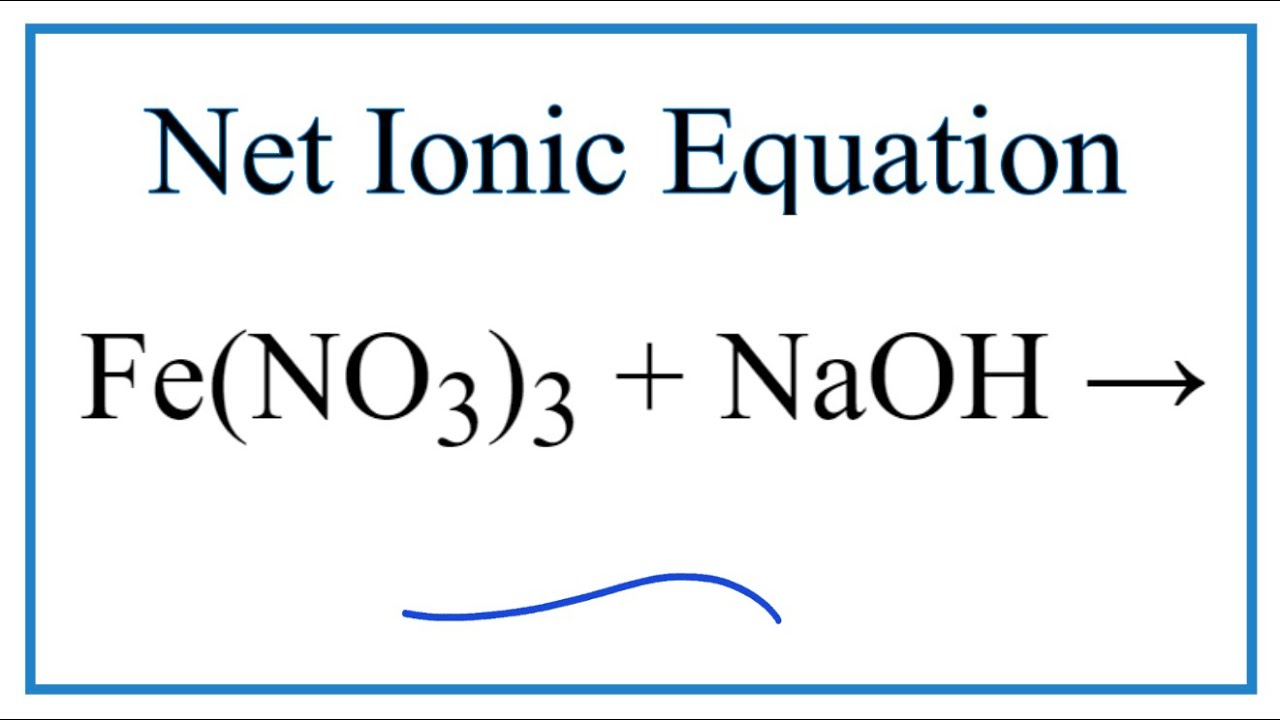SOLVED: A reaction of Aluminum metal with Iron nitrate will produce Iron metal and Aluminum nitrate as shown; Fe(NOslziaql Ala) Ferl AI(NOs) (unbalanced) 1.898 of Al reacts with 68.0 mL of 1.15
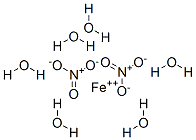
![7782-61-8・Iron(III) Nitrate Nonahydrate, 99.9%・097-02812・091-02815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation 7782-61-8・Iron(III) Nitrate Nonahydrate, 99.9%・097-02812・091-02815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/sc/01/7782-61-8.png)
7782-61-8・Iron(III) Nitrate Nonahydrate, 99.9%・097-02812・091-02815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation






![Solved 17. Iron(II) nitrate [Fe(NO3)2 (aq)] reacts with | Chegg.com Solved 17. Iron(II) nitrate [Fe(NO3)2 (aq)] reacts with | Chegg.com](https://media.cheggcdn.com/media/64b/64b7e906-8e0c-43f2-97f5-f1ae230f5ba3/phpv01T0v.png)