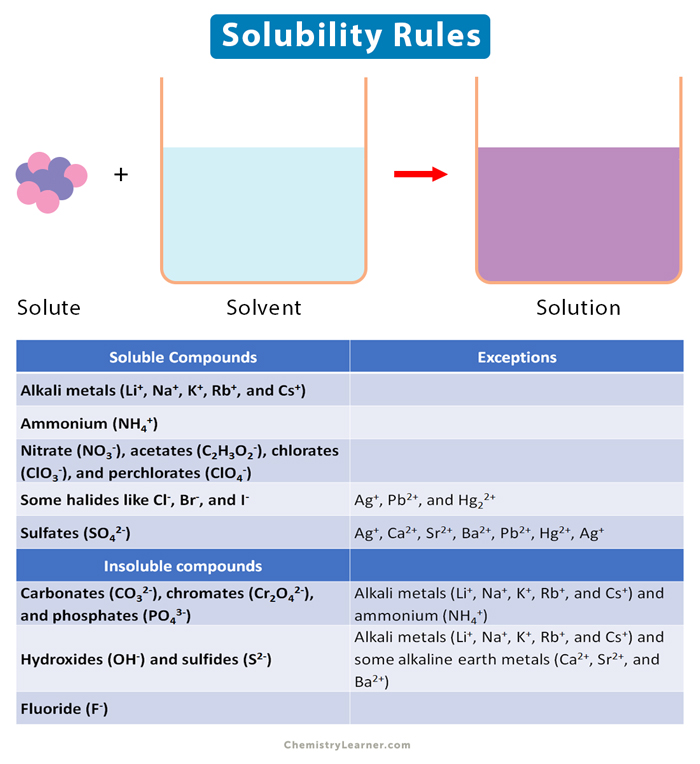
SOLVED: a) Determine the molar solubility of iron(II) fluoride in water Iron (II) fluoride Ksp 2.36 x 10-6 b) The iron(II) fluoride was then treated with a 2.50 M solution of KSCN:

Lithium Insertion Mechanism in Iron Fluoride Nanoparticles Prepared by Catalytic Decomposition of Fluoropolymer | ACS Applied Energy Materials

What is the molar solubility of iron (III) hydroxide (Ksp = 1.6 x 10-39) in a 0.25 M solution of Fe(NO3) 3? - Quora

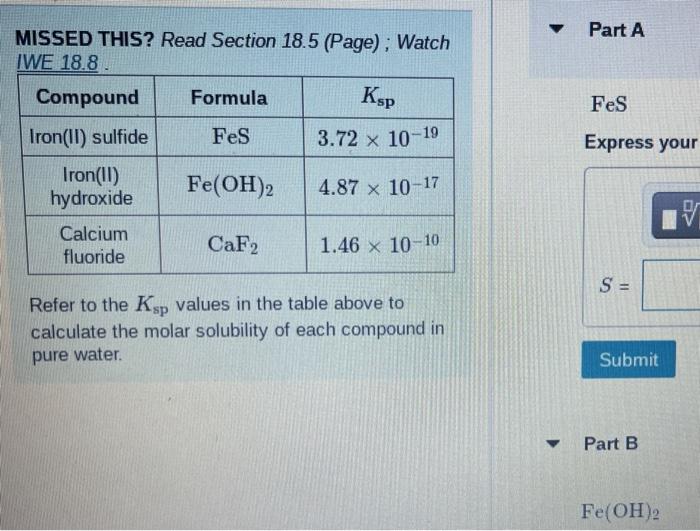
SOLVED: Compound Formula Ksp Iron(II) sulfide FeS 3.72 X 10-19 Magnesium Mg(OH)2 2.06 X 10-13 hydroxide Calcium CaF2 1.46 X 10-10 fluoride Refer to the Ksp values in the table above to

SOLVED: Here is some information on four solid substances: Substance Solubility in watcr Calcium fluoride, CaF: Potassium nitrate, KNO: Sodium sulfate, NaSO. insoluble soluble soluble Iron, Fe insoluble Can the following pairs

Synthesis and Reactivity of Low-Coordinate Iron(II) Fluoride Complexes and Their Use in the Catalytic Hydrodefluorination of Fluorocarbons | Journal of the American Chemical Society
![SOLVED: a) Determine the molar solubility of iron(II) fluoride in water b) The iron(II) fluoride was then treated with a 2.50 M solution of KSCN: Using the formation constant Kform for [FeSCN]+ SOLVED: a) Determine the molar solubility of iron(II) fluoride in water b) The iron(II) fluoride was then treated with a 2.50 M solution of KSCN: Using the formation constant Kform for [FeSCN]+](https://cdn.numerade.com/ask_images/8c298f44a68d49419533742c946117f5.jpg)