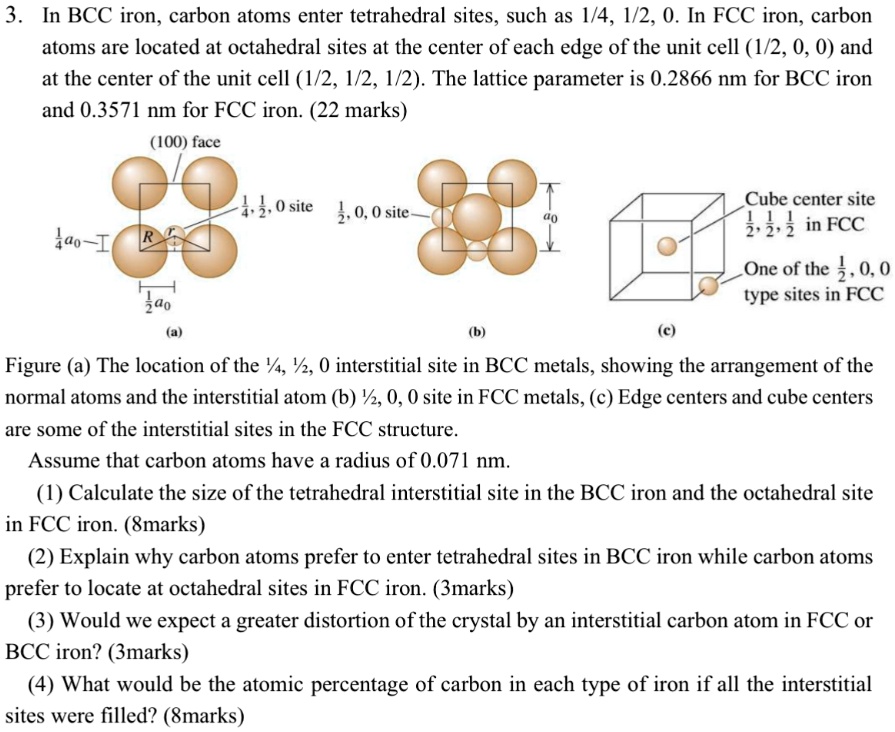
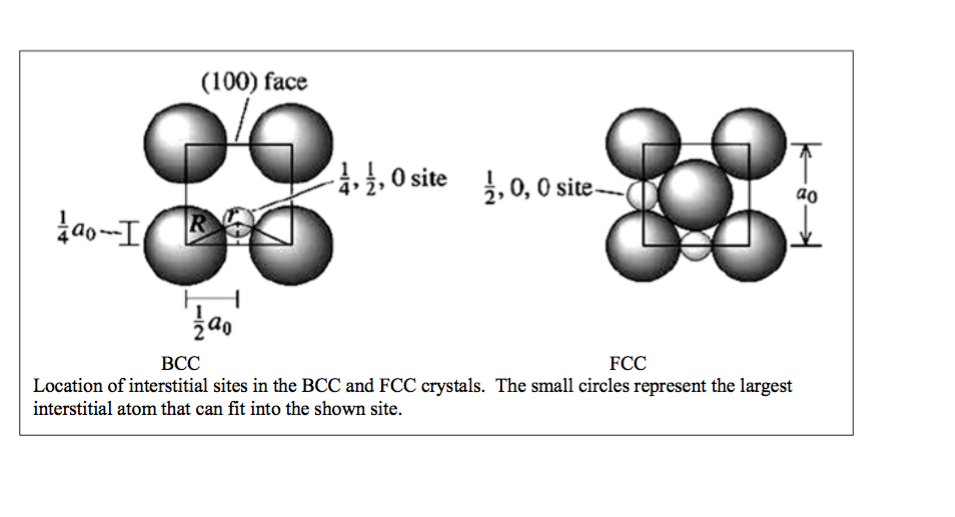
SOLVED: 3 In BCC iron, carbon atoms enter tetrahedral sites, such as 1/4, 1/2, 0. In FCC iron, carbon atoms are located at octahedral sites at the center of each edge of

γ-Iron Phase Stabilized at Room Temperature by Thermally Processed Graphene Oxide | Journal of the American Chemical Society
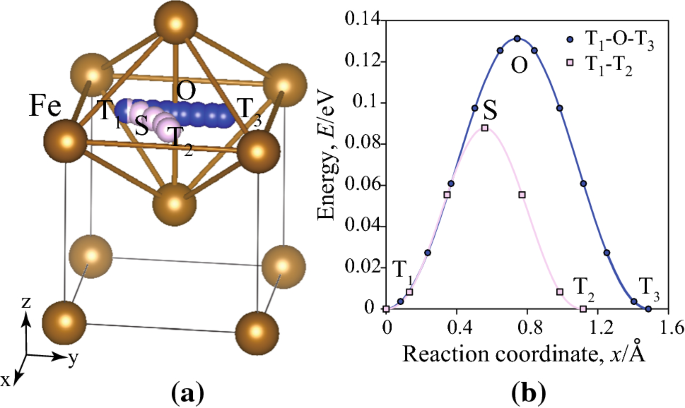
Point defect interactions in iron lattice: a first-principles study - RSC Advances (RSC Publishing) DOI:10.1039/C6RA05969D

FCC, BCC and BCT lattice system of different forms of iron with carbon... | Download Scientific Diagram

Schematics of ͑ a ͒ fcc-Pd ͑ 110 ͒ , ͑ b ͒ bcc-Fe ͑ 110 ͒ , and ͑ c ͒... | Download Scientific Diagram

Volume Change During BCC to FCC Iron transformation//Calculation of Volume change in transformations - YouTube

In FCC iron, carbon atoms are located at octahedral sites at the center of each edge of the unit cell (1/2,0,0) and at the center of the unit cell (1/2,1/2,1/2) . Illustrate
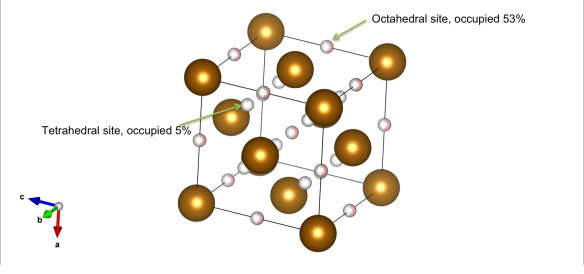
What Is the Difference Between FCC and BCC? (Crystal Structure, Properties, Interstitial Sites, and Examples) – Materials Science & Engineering

SOLVED: The lattice parameter (aFcc) 3.571 x 10/0 m for FCC iron; (aecc) 2.866 X 10-10 m for BCC iron; Radius of Iron atom (rre) 1.241 X 1010 mj Radius of Carbon atom (rc) 0.71 x 1010 mj






![Crystallography of Iron Films [IAP/TU Wien] Crystallography of Iron Films [IAP/TU Wien]](https://www.iap.tuwien.ac.at/www/_media/surface/stm_gallery/bcc110vs_square.gif)




![Crystallography of Iron Films [IAP/TU Wien] Crystallography of Iron Films [IAP/TU Wien]](https://www.iap.tuwien.ac.at/www/_media/surface/stm_gallery/fcc_bcc.gif)