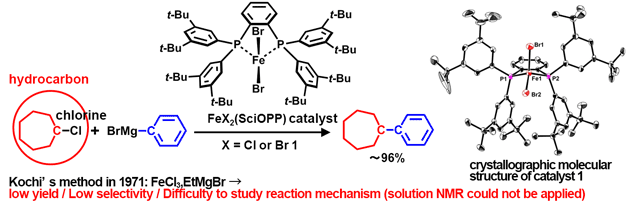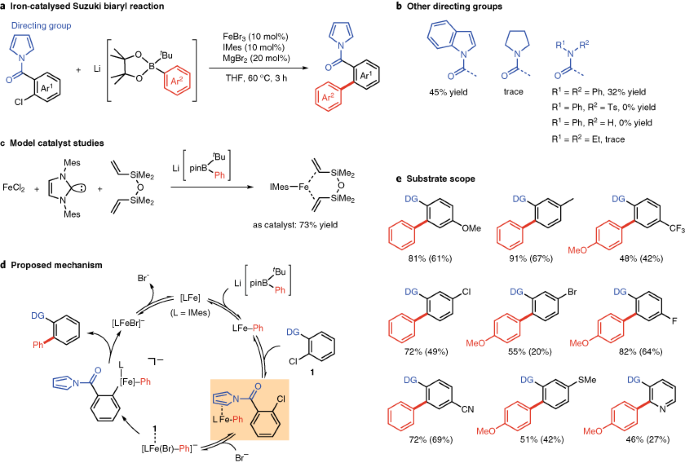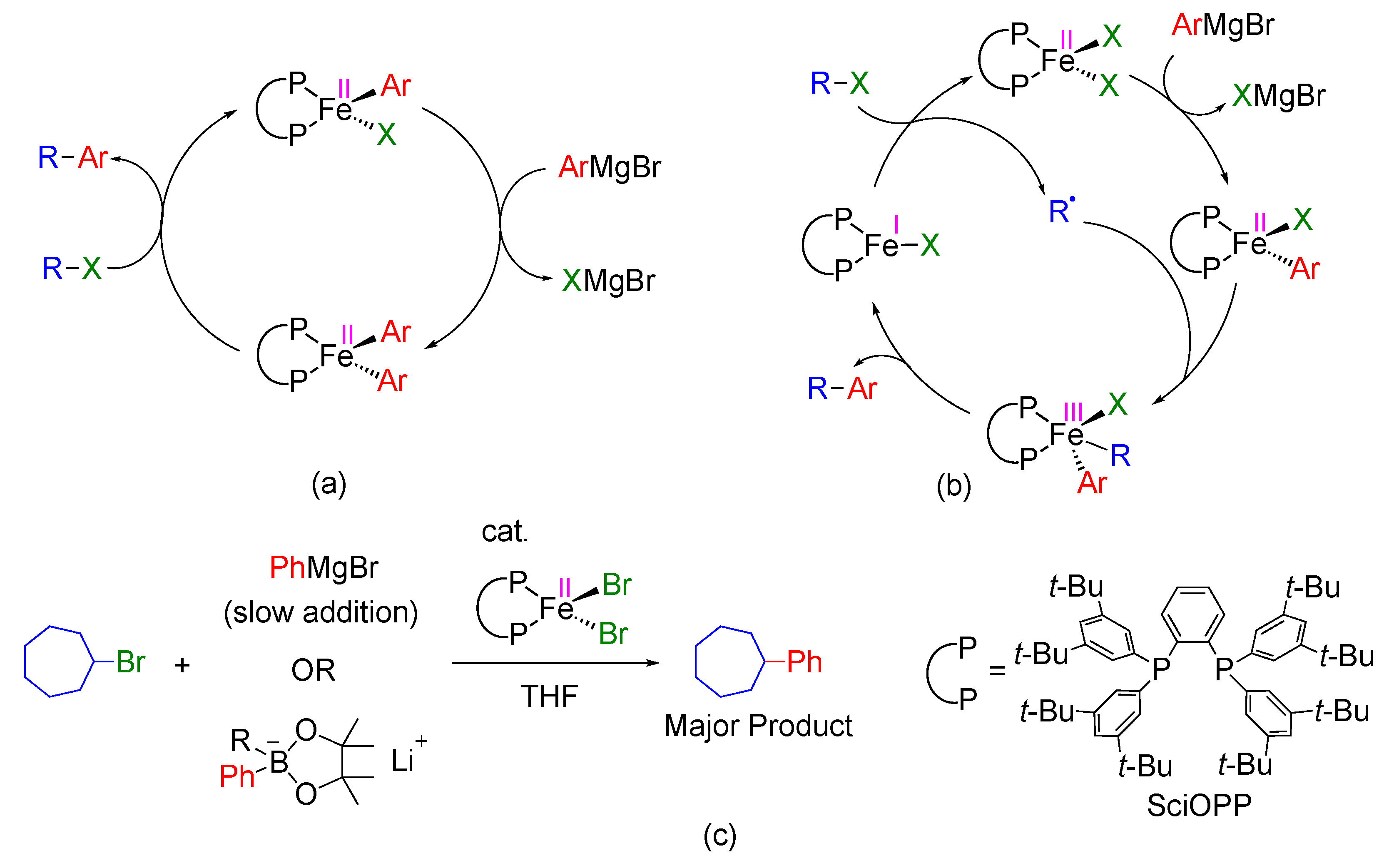
Molecules | Free Full-Text | A DFT Study on FeI/FeII/FeIII Mechanism of the Cross-Coupling between Haloalkane and Aryl Grignard Reagent Catalyzed by Iron-SciOPP Complexes

Iron-Catalyzed Suzuki–Miyaura Cross-Coupling Reactions between Alkyl Halides and Unactivated Arylboronic Esters | Organic Letters

Table 2 from Iron(ii)-catalyzed sulfur directed C(sp3)-H bond amination/C-S cross coupling reaction. | Semantic Scholar

Iron-Catalyzed Cross-Coupling of Primary and Secondary Alkyl Halides with Aryl Grignard Reagents | Journal of the American Chemical Society
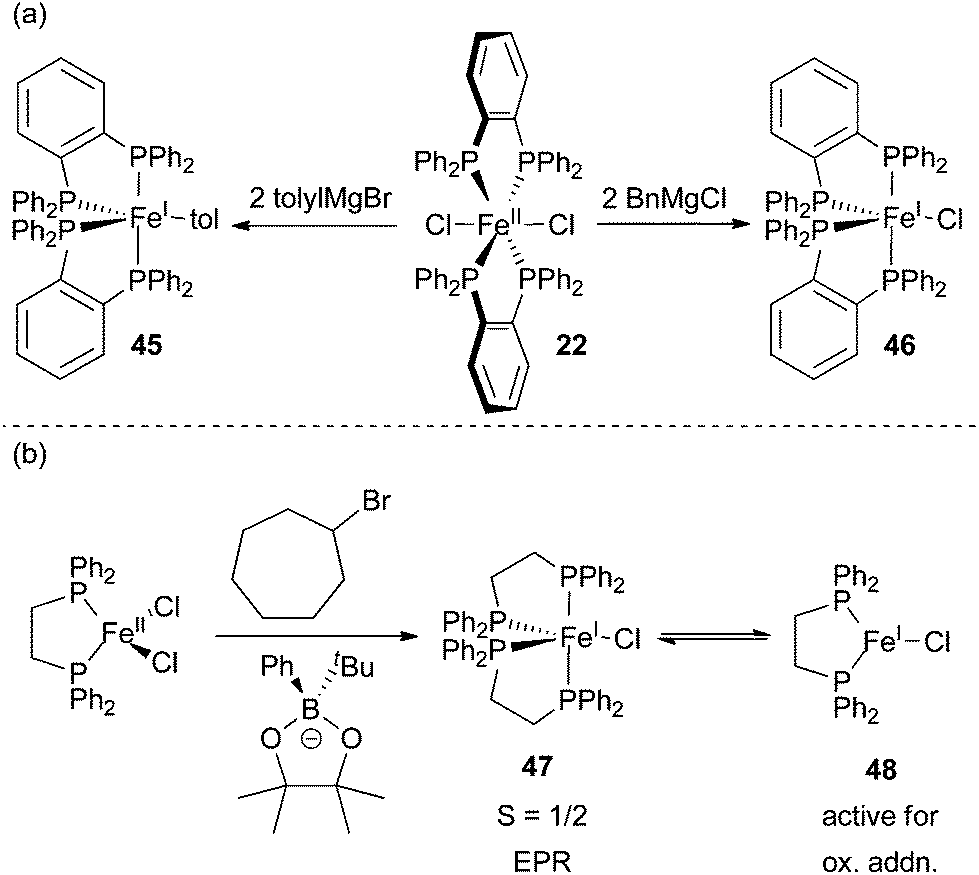
Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H

Intermediates and Mechanism in Iron-Catalyzed Cross-Coupling | Journal of the American Chemical Society

Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H

Iron-Catalyzed Cross-Coupling of Thioesters and Organomanganese Reagents | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H
![Iron Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | TCI Chemicals (India) Pvt. Ltd. Iron Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | TCI Chemicals (India) Pvt. Ltd.](https://www.tcichemicals.com/assets/cms-images/S012-Fe-1.gif)
Iron Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | TCI Chemicals (India) Pvt. Ltd.
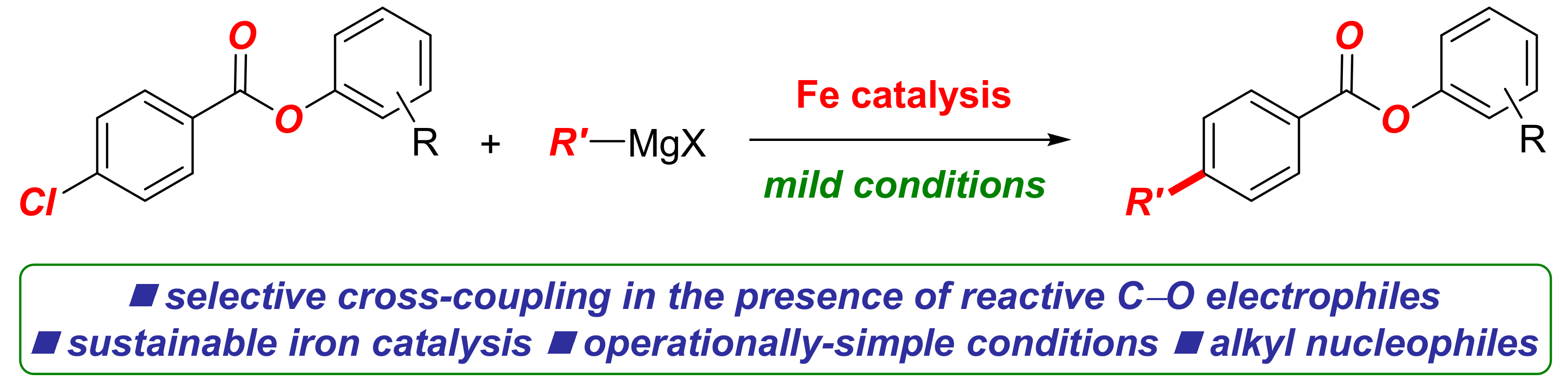
Molecules | Free Full-Text | Iron-Catalyzed C(sp2)–C(sp3) Cross-Coupling of Aryl Chlorobenzoates with Alkyl Grignard Reagents

Iron-Catalyzed Suzuki–Miyaura Cross-Coupling Reactions between Alkyl Halides and Unactivated Arylboronic Esters | Organic Letters

Iron‐Catalyzed Cross‐Couplings in the Synthesis of Pharmaceuticals: In Pursuit of Sustainability - Piontek - 2018 - Angewandte Chemie International Edition - Wiley Online Library

Iron‐Catalyzed Cross‐Couplings in the Synthesis of Pharmaceuticals: In Pursuit of Sustainability - Piontek - 2018 - Angewandte Chemie International Edition - Wiley Online Library
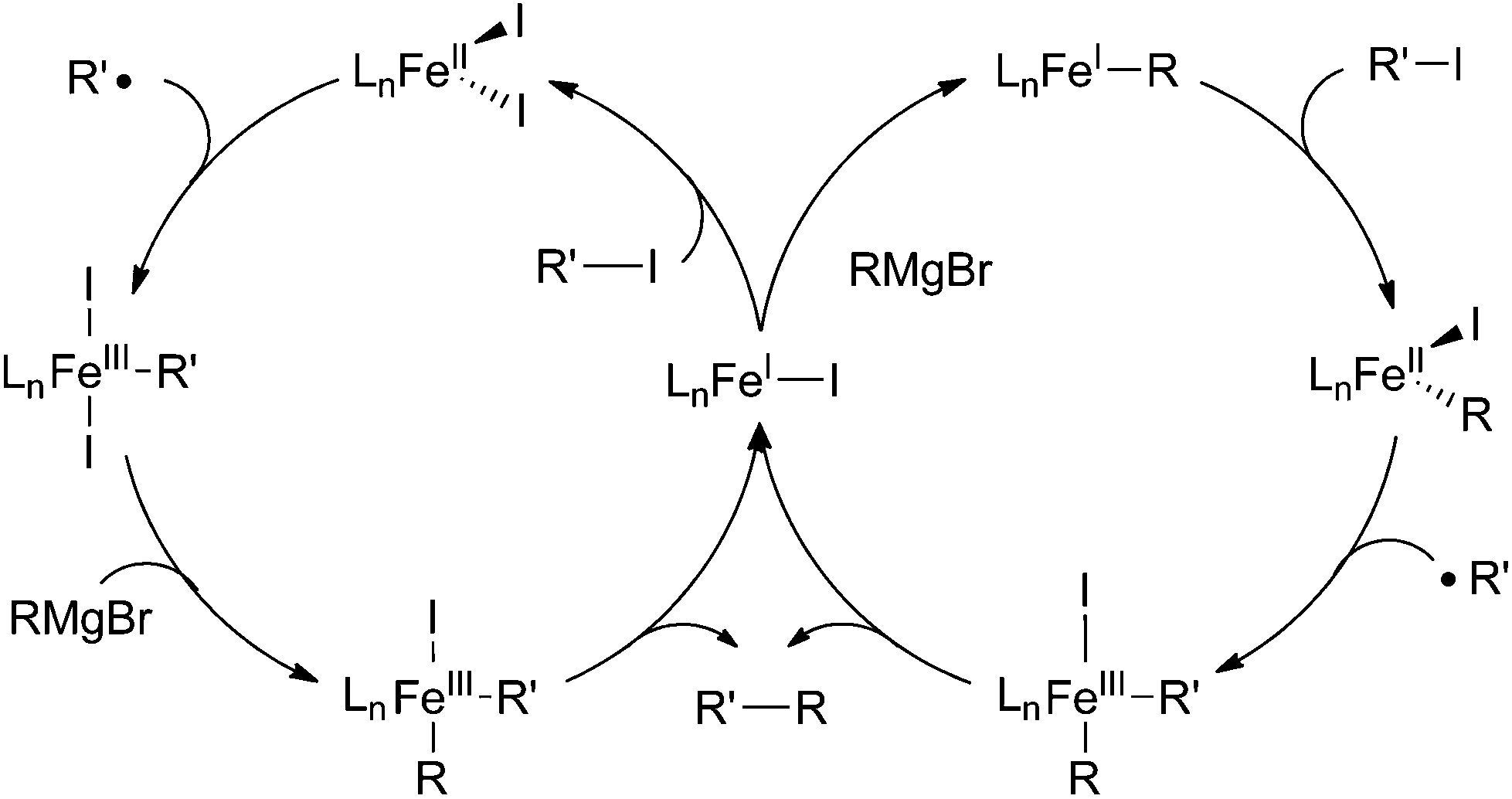
Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H

N-Butylpyrrolidone (NBP) as a non-toxic substitute for NMP in iron-catalyzed C(sp2)–C(sp3) cross-coupling of aryl chlorides | Green Chemistry International