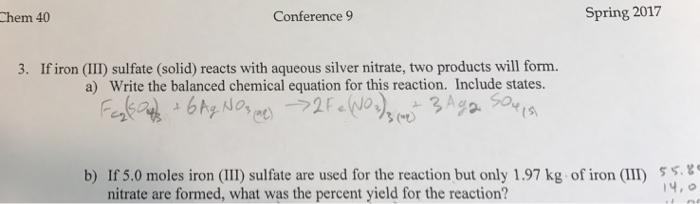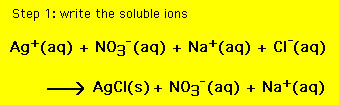
SOLVED: According to the following reaction, how many grams of silver nitrate are required for the complete reaction of 26.9 grams of iron(II) chloride ? iron(II) chloride(aq) + silver nitrate(aq) iron(II) nitrate(aq) +
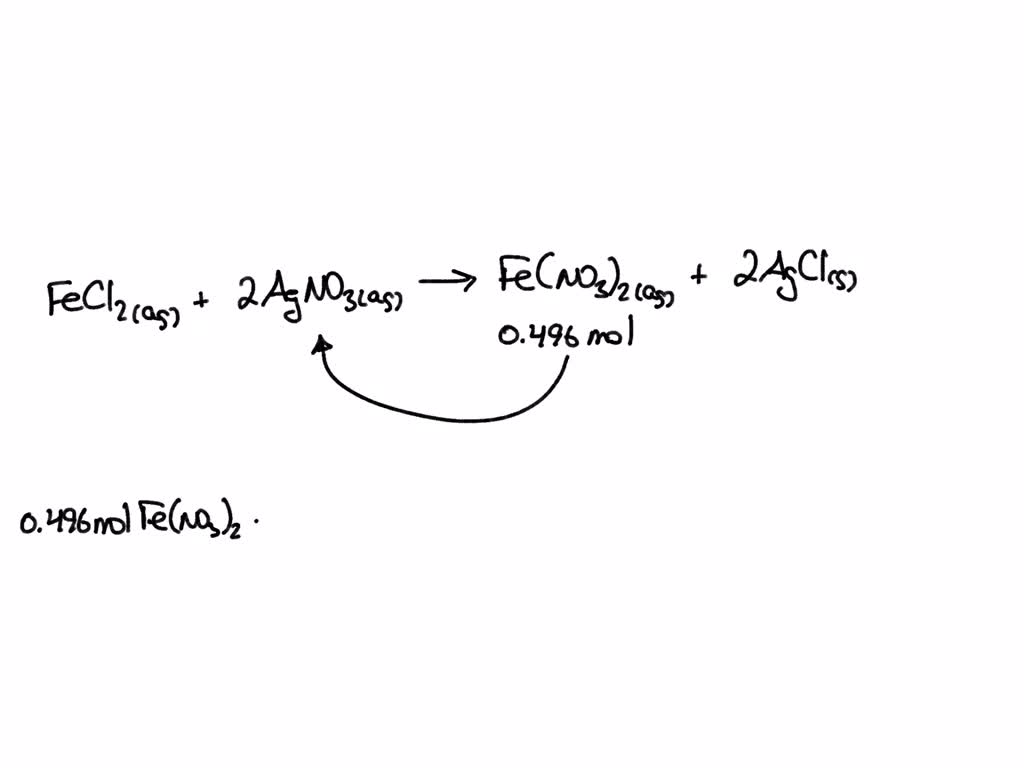
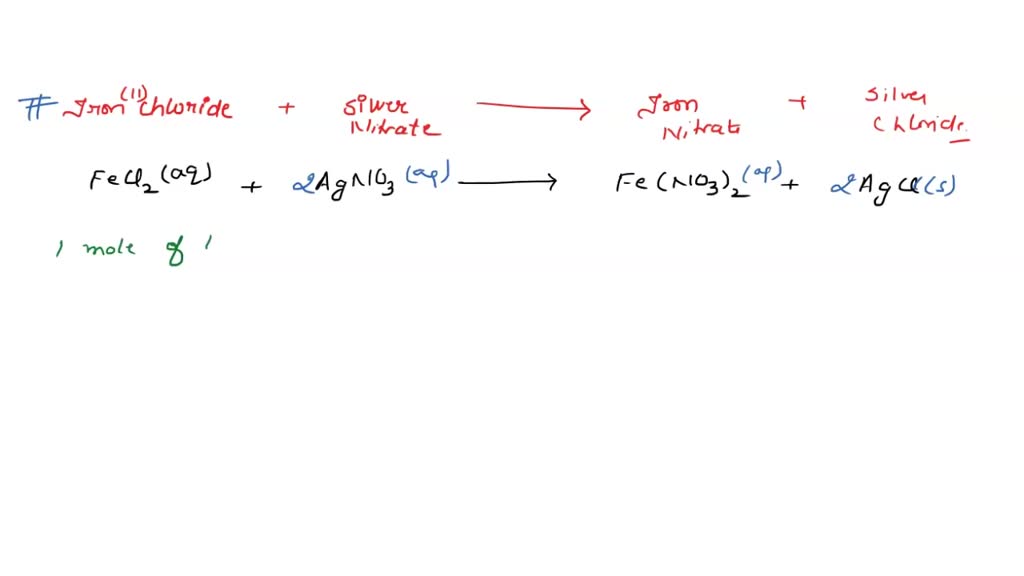
SOLVED: Silver nitrate reacts with iron (III) chloride to produce silver chloride and iron (III) nitrate. In a particular experiment, a solution containing 25.0 g of silver nitrate is completely reacted. Write

SOLVED: The following molecular equation represents the reaction that occurs when aqueous solutions of silver([) nitrate and iron(III) bromide are combined. 3AgNO: (aq) + FeBrs (aq) 3AgBr(s) + Fe(NO:)3 (aq) Write the

Fe+AgNO3=Fe(NO3)2+Ag Balanced Equation||Iron+Silver nitrate=Iron(ii)nitrate+ Silver Balanced Equation - YouTube

SOLVED: 41, Silver nitrate, AgNO3(aq) , reacts with iron(III) chloride, FeCl;(aq), to produce silver chloride; 'AgCl(s), and iron(III) nitrate; Fe(NOz)3(aq) 3AgNOs(aq) - FeClz(aq) 3AgCl(s) + Fe(NOs)s(aq) a. Ifa solution containing 18.00 g