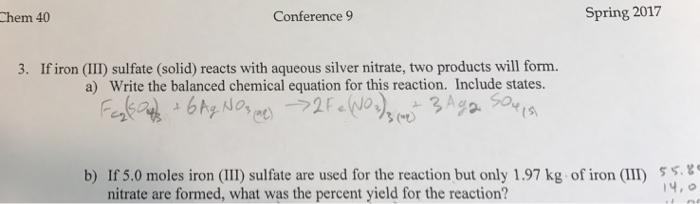SOLVED: The following molecular equation represents the reaction that occurs when aqueous solutions of silver([) nitrate and iron(III) bromide are combined. 3AgNO: (aq) + FeBrs (aq) 3AgBr(s) + Fe(NO:)3 (aq) Write the

OneClass: One way the U.S. Environmental Protection Agency (EPA) tests for chloride contaminants in ...

An iron nail is dipped in a silver nitrate solution. After some time, the solution turns green due to the formation of iron nitrate and silver. Which of the following equations represents

What happens when iron nail is placed in silver nitrate solution? (ii)iron strip is dipped in zinc sulphate solution?

SOLVED: 41, Silver nitrate, AgNO3(aq) , reacts with iron(III) chloride, FeCl;(aq), to produce silver chloride; 'AgCl(s), and iron(III) nitrate; Fe(NOz)3(aq) 3AgNOs(aq) - FeClz(aq) 3AgCl(s) + Fe(NOs)s(aq) a. Ifa solution containing 18.00 g
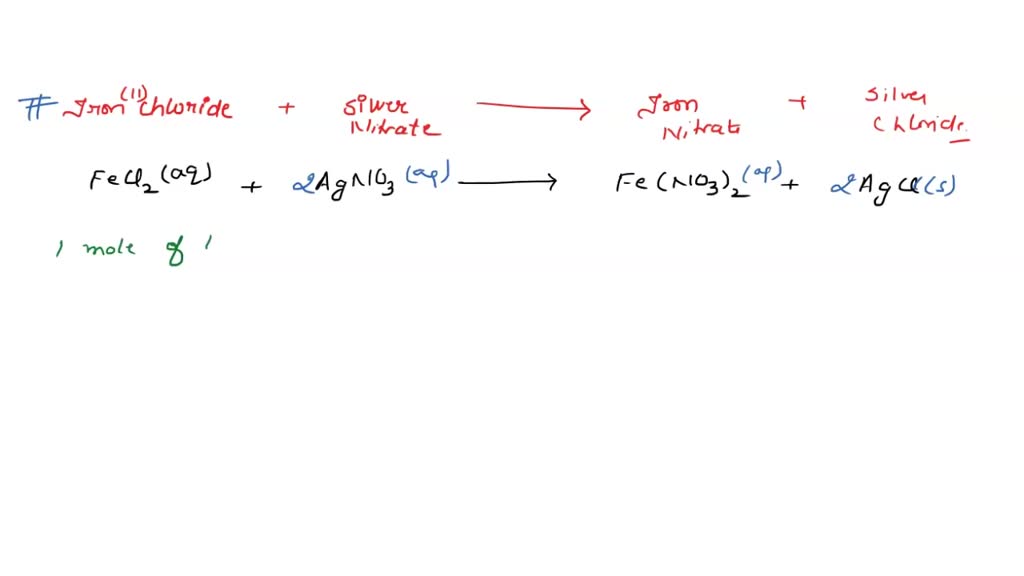
SOLVED: According to the following reaction, how many grams of silver nitrate are required for the complete reaction of 26.9 grams of iron(II) chloride ? iron(II) chloride(aq) + silver nitrate(aq) iron(II) nitrate(aq) +

Schematic representation for the synthesis of silver nitrate (AgNO 3 ),... | Download Scientific Diagram

inorganic chemistry - Is an iron (II) sulphate solution supposed to turn cloudy when silver nitrate is added? - Chemistry Stack Exchange

An element X displaces iron from the aqueous solution of iron sulphate .List your observations if the - Brainly.in