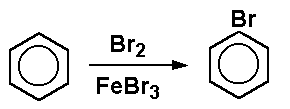
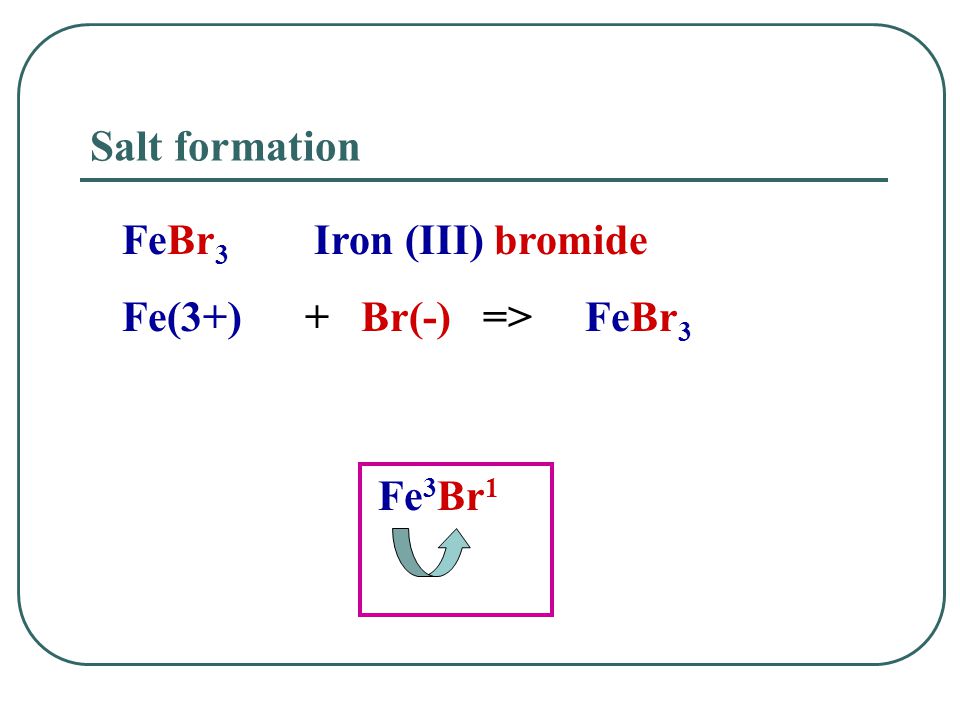
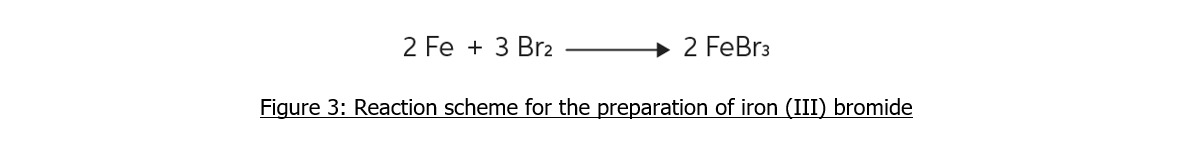
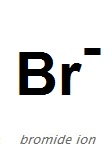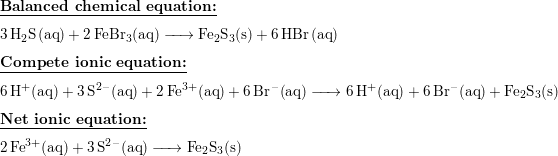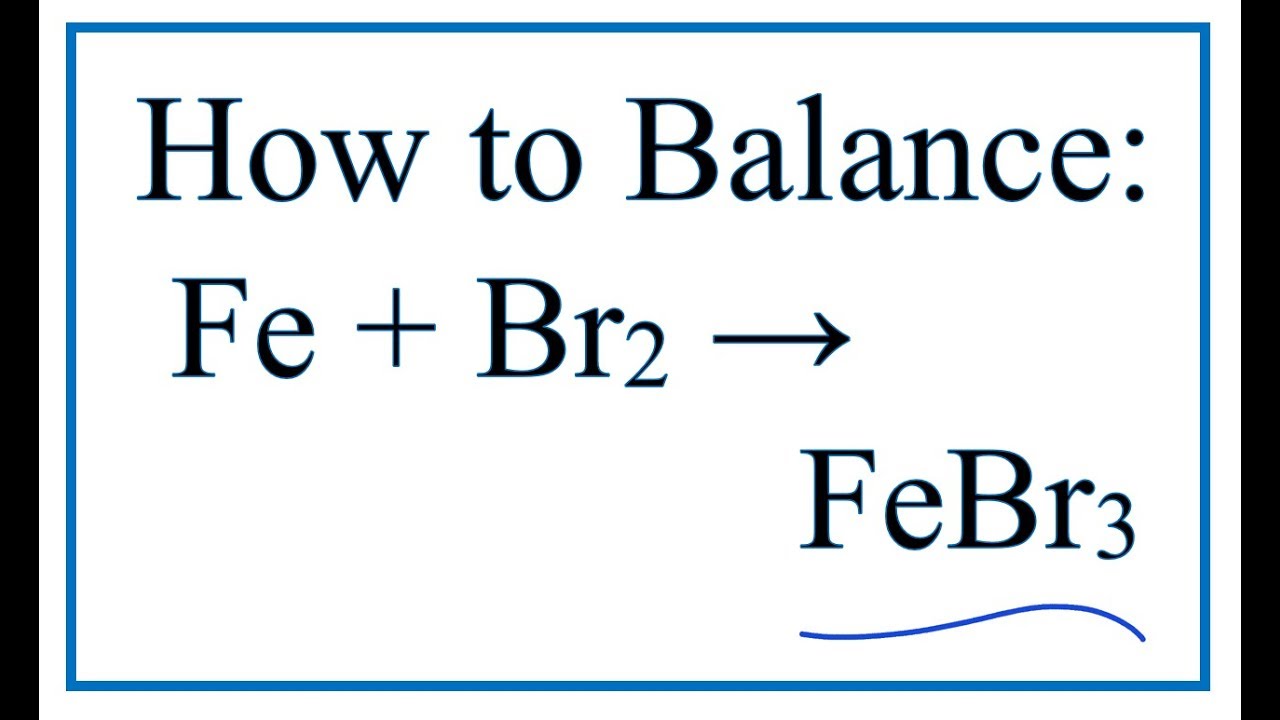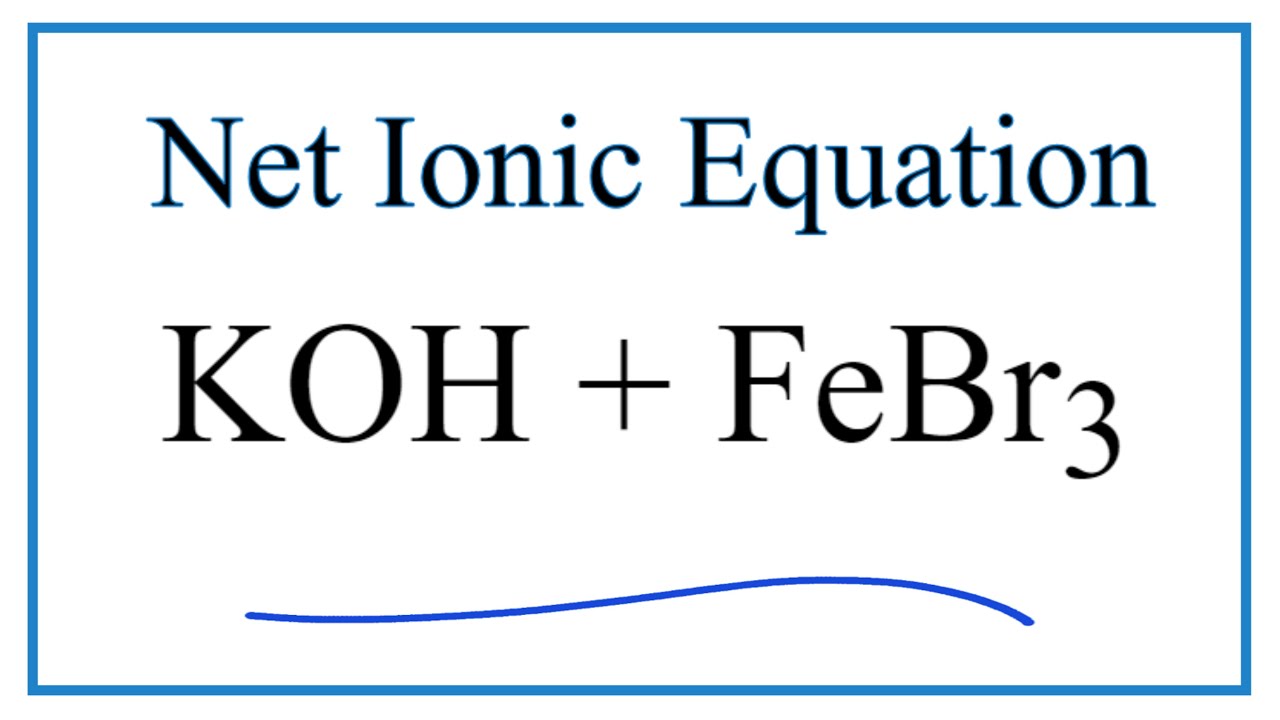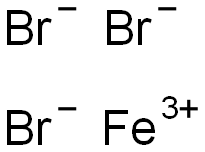Iron(III)Bromide - Iron(III)Bromide,Cupric Salts,Xanthan Gum,CMC,Citric Acid,TCCA,Water Treatment Chemicals Supplier,Food Additives Supplier-Sunvel Chemical Co.,Ltd
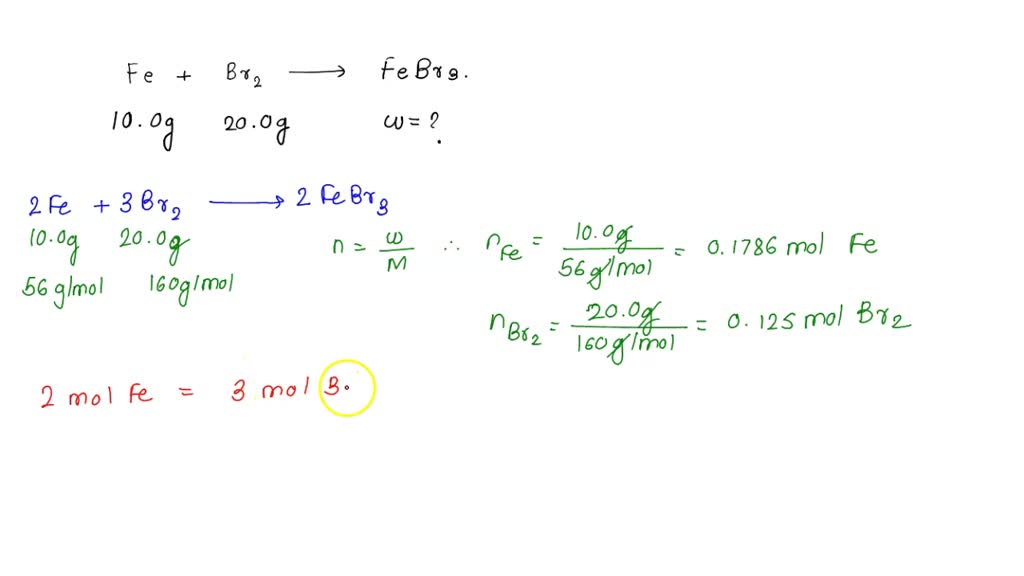
SOLVED: Iron can react with diatomic bromine to form iron(III) bromide. If a 10.0 g lump of iron were placed in 20.0 g of liquid bromine, what mass of iron(III) bromide would

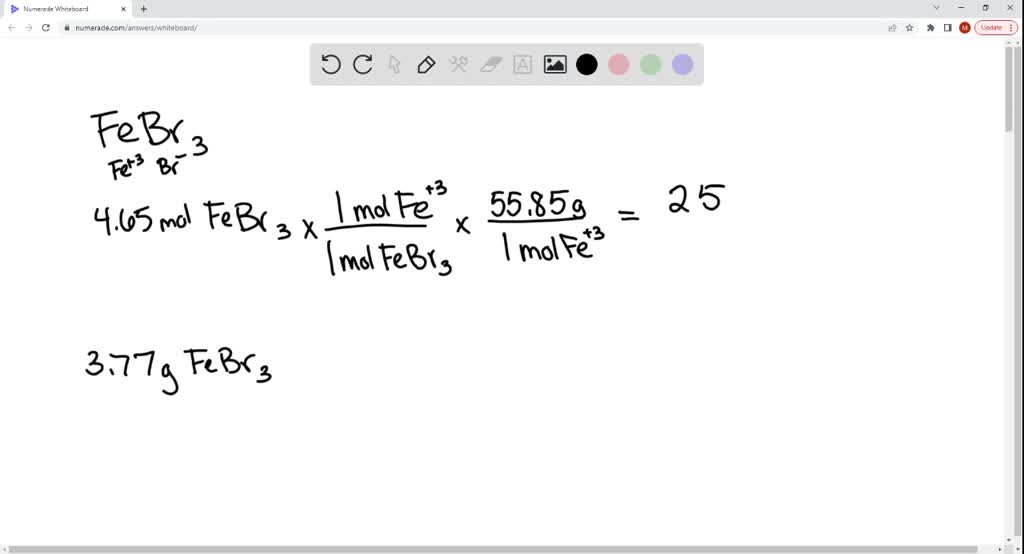
SOLVED: The following molecular equation represents the reaction that occurs when aqueous solutions of silver([) nitrate and iron(III) bromide are combined. 3AgNO: (aq) + FeBrs (aq) 3AgBr(s) + Fe(NO:)3 (aq) Write the