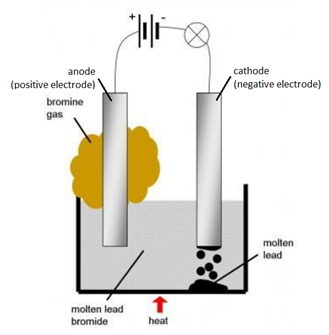
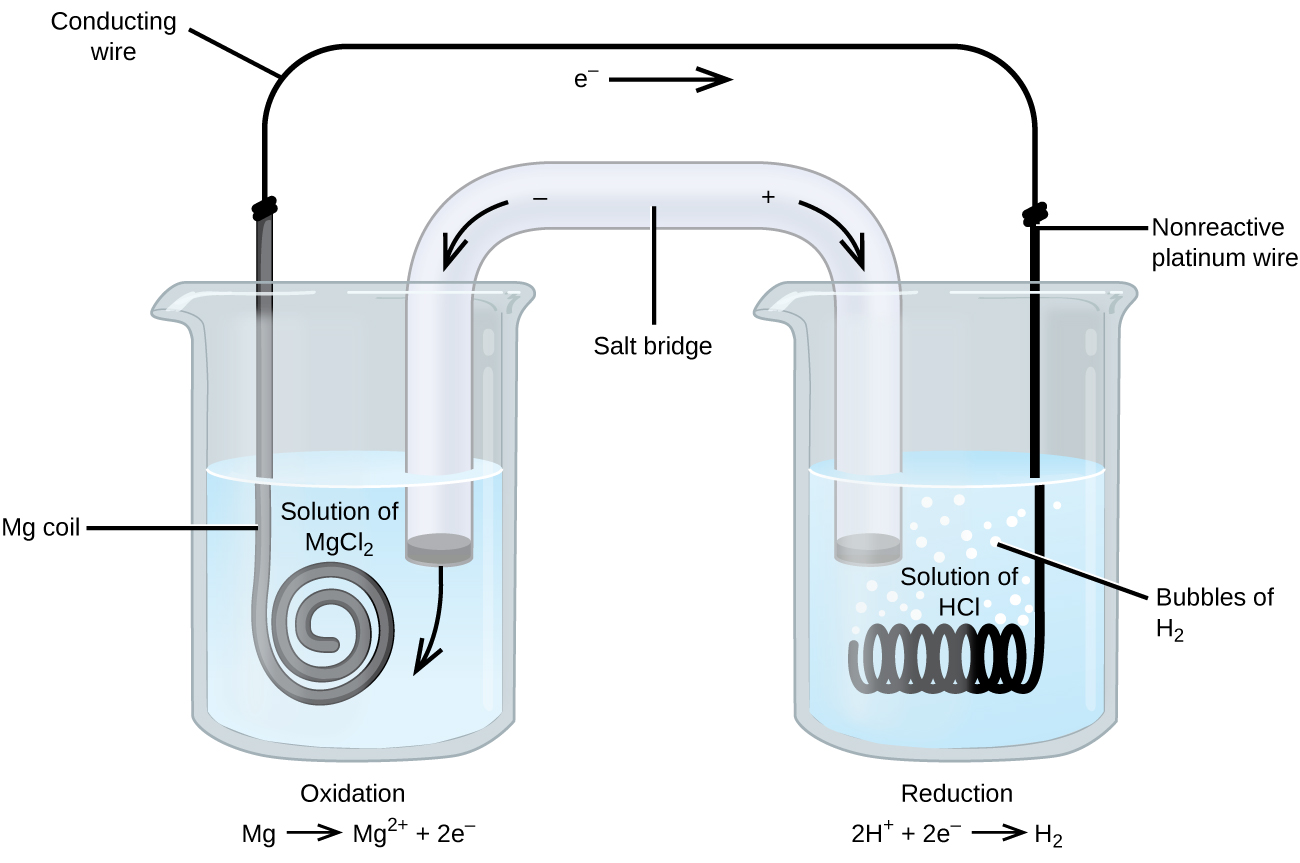
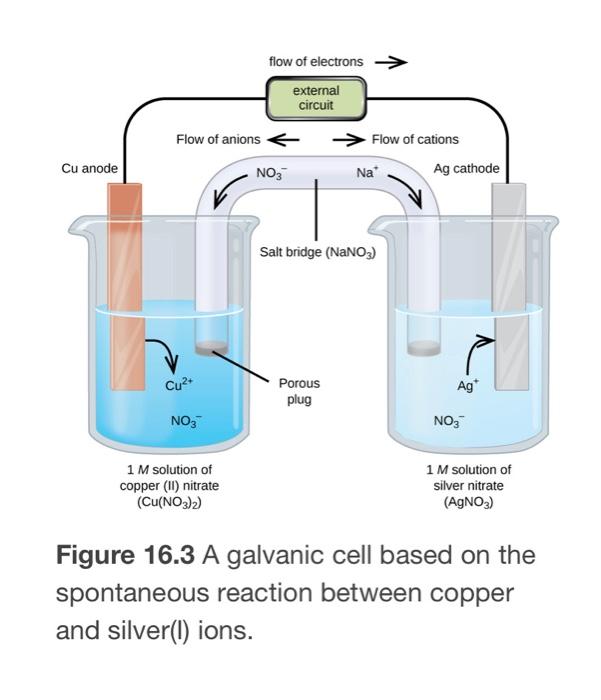
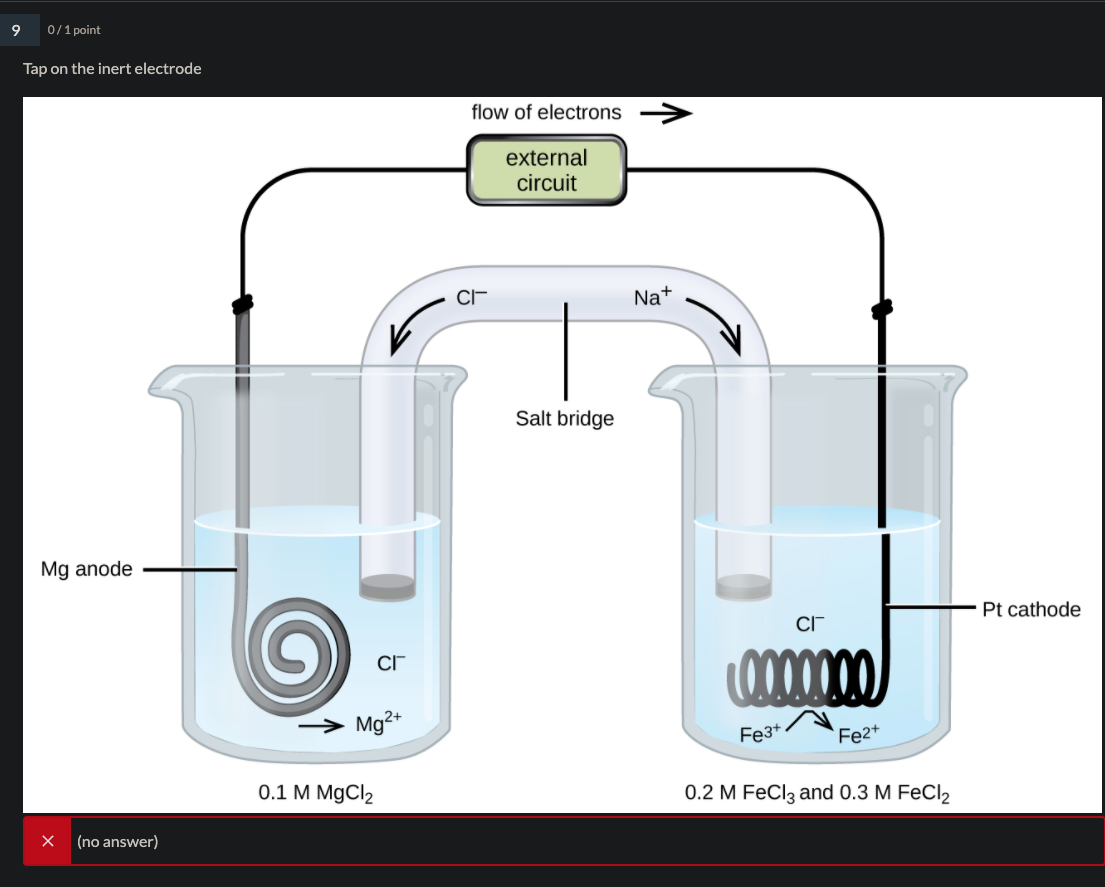
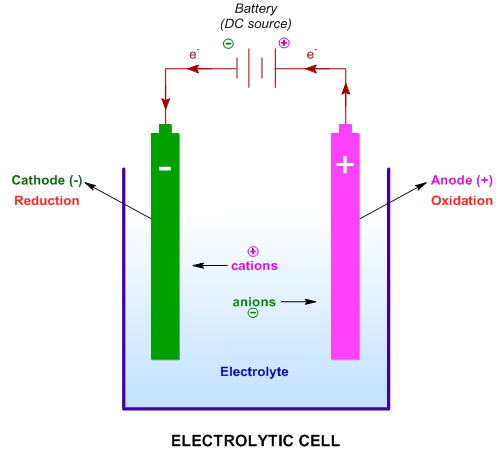
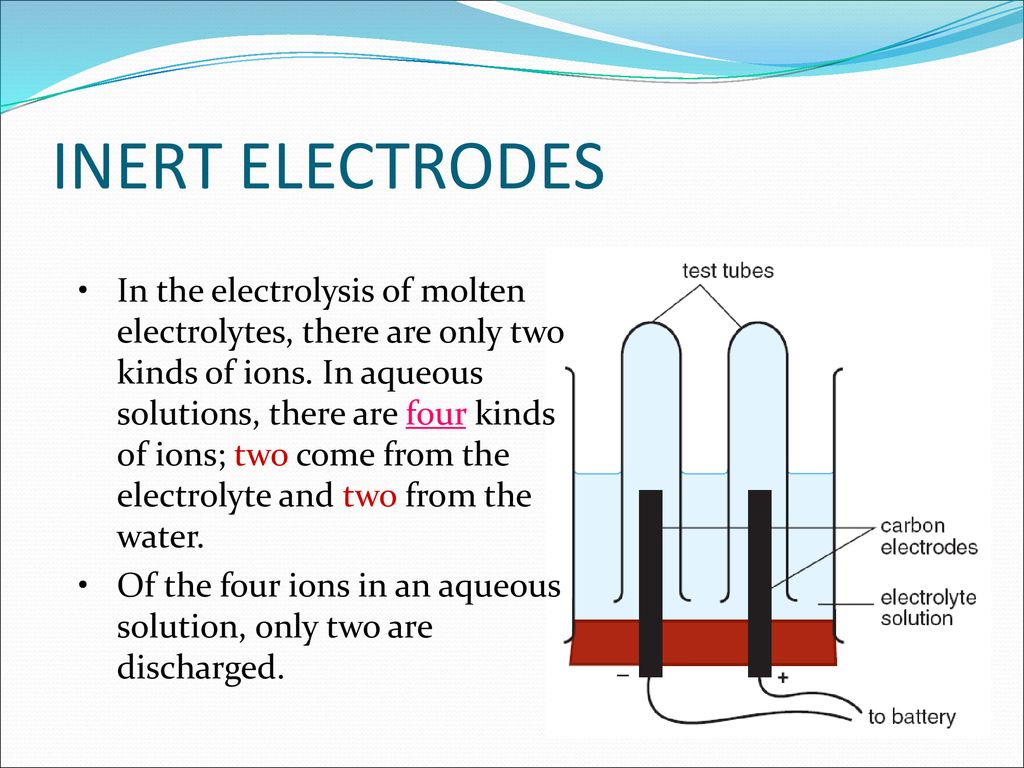
In reference below, for the electrolysis of molten calcium chloride with inert electrodes. The following observations are made when current is supplied: (i) Bubbles of pale green chlorine gas, Cl_2, are produced

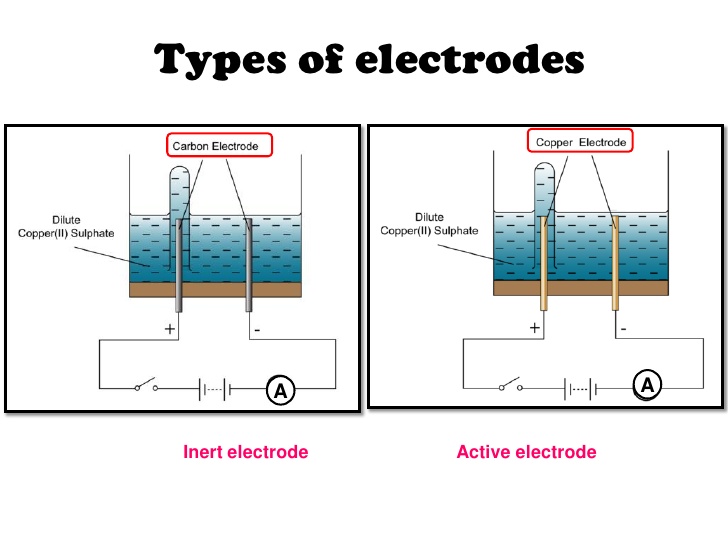
electrochemistry - Would a voltaic cell still work if you replaced both the cathode and anode with a carbon/graphite electrode? - Chemistry Stack Exchange
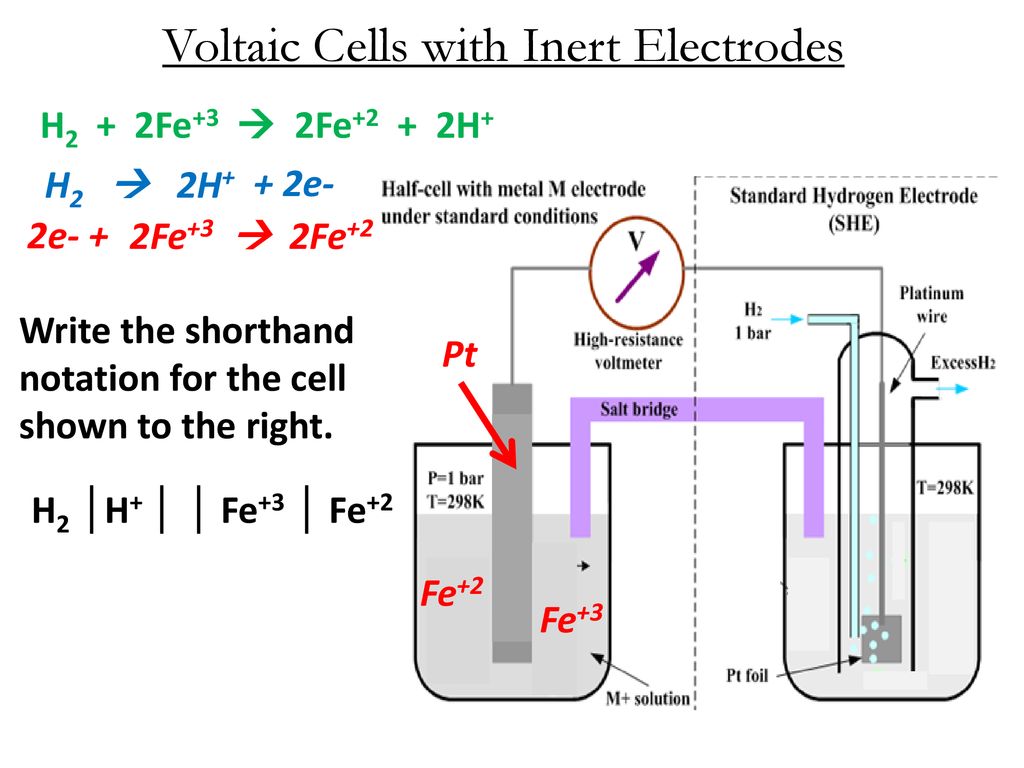
Quiz (Two Half Cells System with Inert Electrodes) 1. A simple chemical cell is shown below: (a) Write a half equation for the