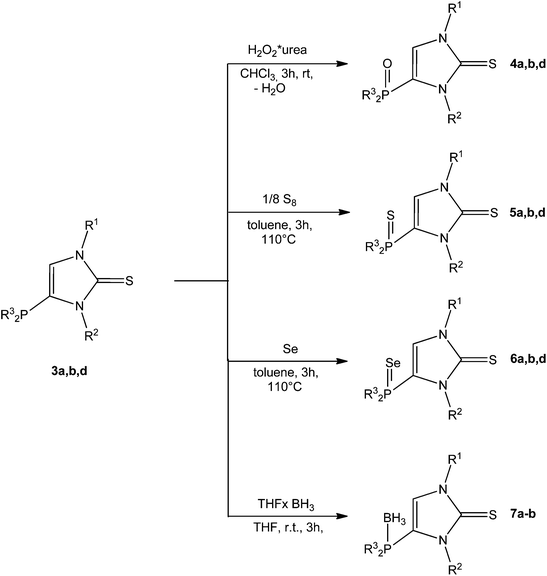
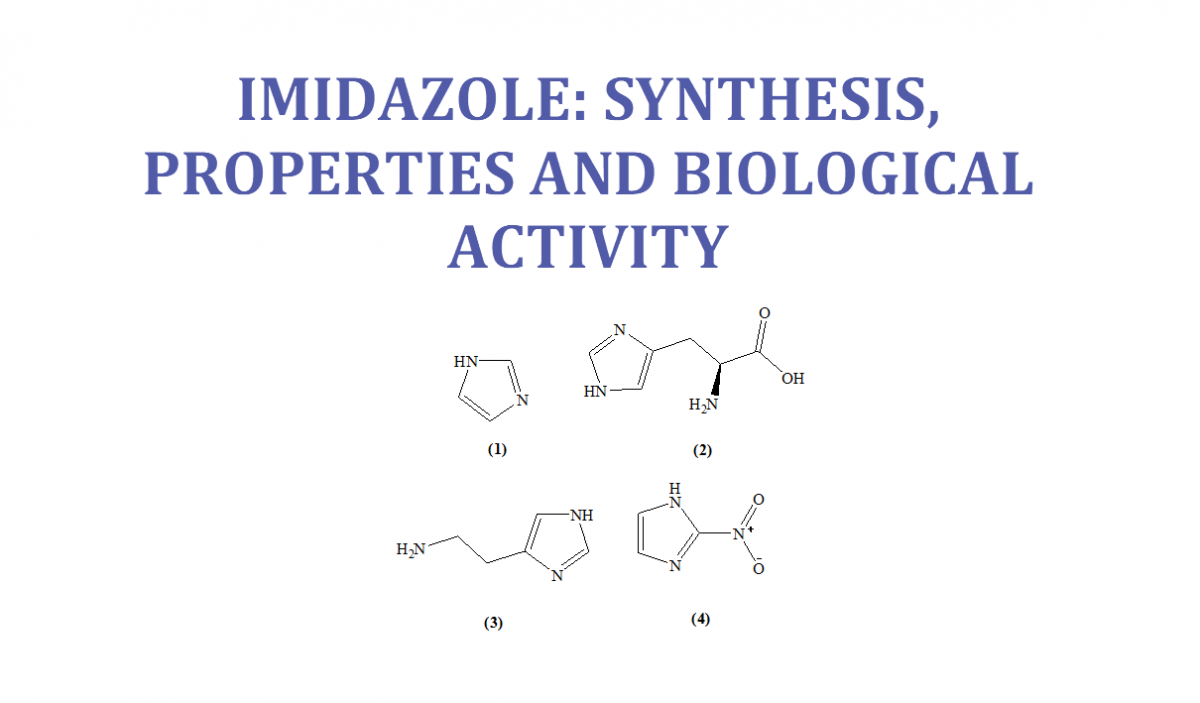
Molecules | Free Full-Text | Imidazole: Synthesis, Functionalization and Physicochemical Properties of a Privileged Structure in Medicinal Chemistry

On the reactivity of imidazole carbamates and ureas and their use as esterification and amidation reagents - ScienceDirect
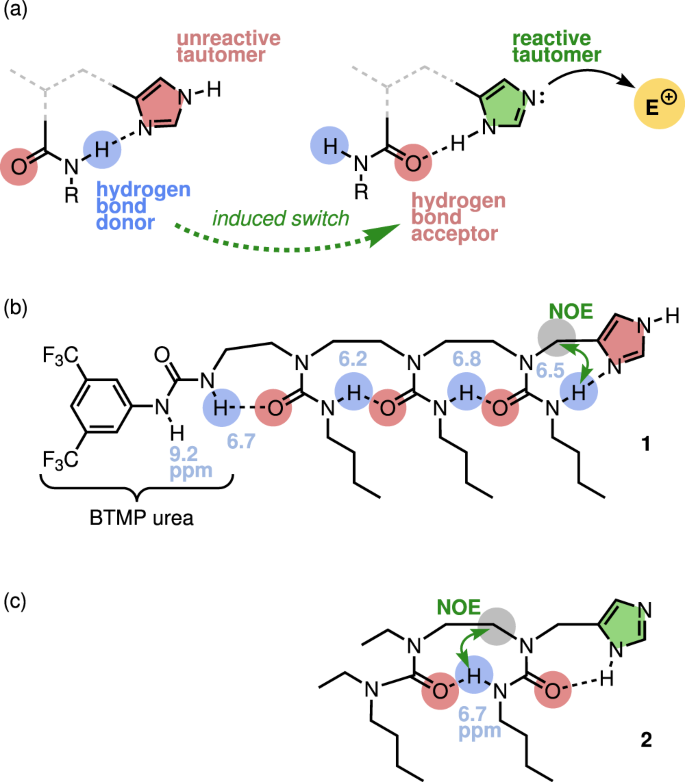
Switching imidazole reactivity by dynamic control of tautomer state in an allosteric foldamer | Nature Communications

Effect of Imidazole and Phenolate Axial Ligands on the Electronic Structure and Reactivity of Oxoiron(IV) Porphyrin π-Cation Radical Complexes: Drastic Increase in Oxo-Transfer and Hydrogen Abstraction Reactivities | Inorganic Chemistry
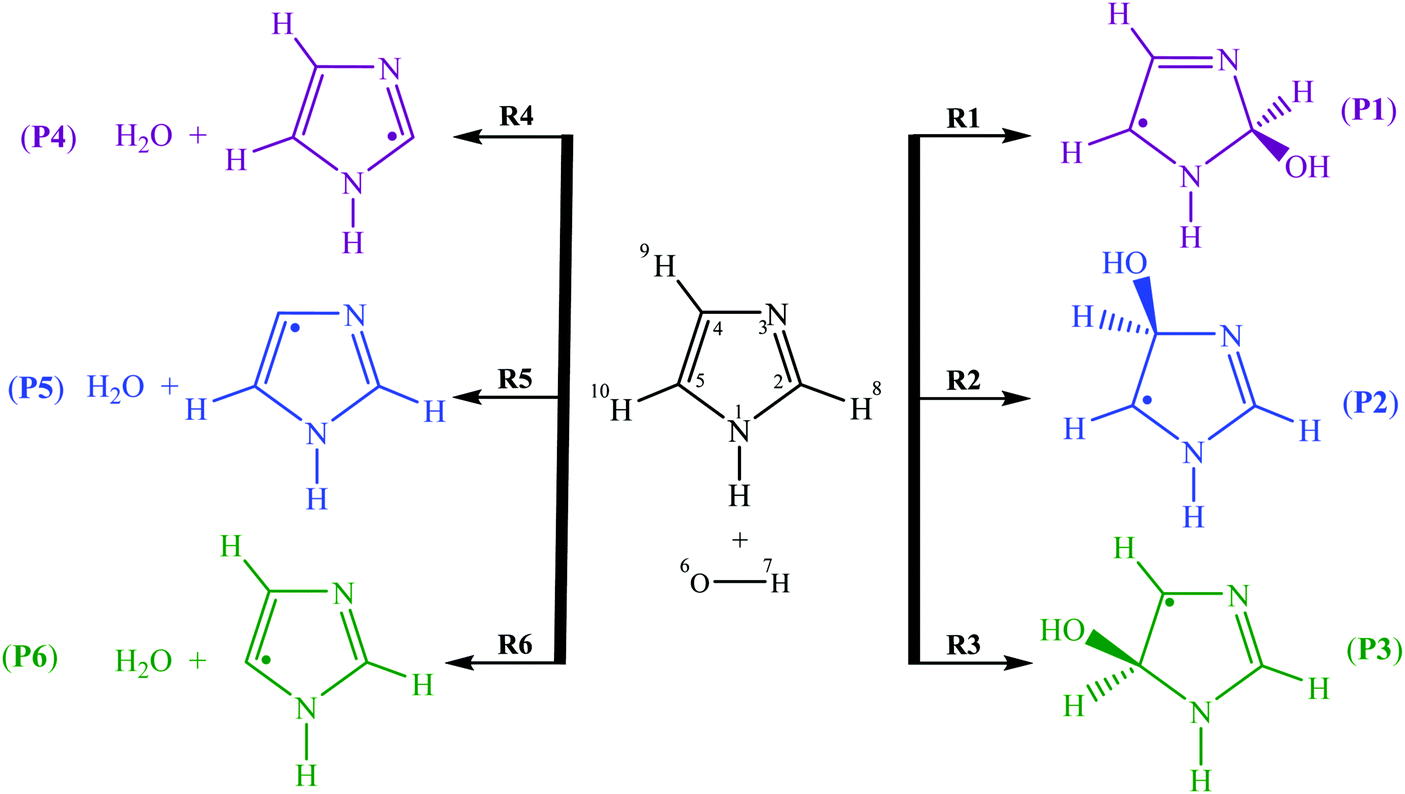
Atmospheric oxidation reactions of imidazole initiated by hydroxyl radicals: kinetics and mechanism of reactions and atmospheric implications - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C9CP00632J

SciELO - Brasil - Are Imidazoles Versatile or Promiscuous in Reactions with Organophosphates? Insights from the Case of Parathion Are Imidazoles Versatile or Promiscuous in Reactions with Organophosphates? Insights from the Case
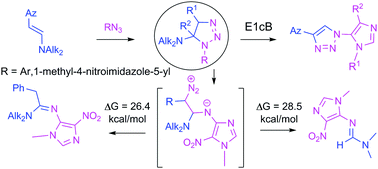
Design and synthesis of imidazoles linearly connected to carbocyclic and heterocyclic rings via a 1,2,3-triazole linker. Reactivity of β-azolyl enamines towards heteroaromatic azides - New Journal of Chemistry (RSC Publishing)

On the reactivity of imidazole carbamates and ureas and their use as esterification and amidation reagents - ScienceDirect