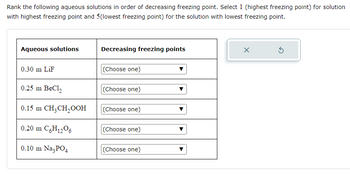
SOLVED: 0.18 m CoCl2 A. Lowest freezing point2. 0.15 m Ba(NO3)2 B. Second lowest freezing point3. 0.26 m NaOH C. Third lowest freezing point4. 0.49 m Sucrose(nonelectrolyte) D. Highest freezing point

Solve this: 2 Which of the following aqueous solution has maximum freezing point (1) 0 01 M - Chemistry - Solutions - 12731753 | Meritnation.com
![Which of the following solutions has higher freezing point? 0.05 M Al2(SO4)3, 0.1 M K3 [ Fe (CN)6 ] Justify. Which of the following solutions has higher freezing point? 0.05 M Al2(SO4)3, 0.1 M K3 [ Fe (CN)6 ] Justify.](https://haygot.s3.amazonaws.com/questions/1309596_1382747_ans_bb0fc6358f864b8eaf6190aeb3485fa3.jpg)
Which of the following solutions has higher freezing point? 0.05 M Al2(SO4)3, 0.1 M K3 [ Fe (CN)6 ] Justify.
7. How to attempt this type of ques. Q.which of the following aqueous solutions has highest freezing point a) 0.1 M NaCl b)0.1M BaCl2 c) 0.1M Al2(SO4)3 d)0.1M urea

Rank the following aqueous solutions from highest to lowest freezing point: 0.1 m FeCl3, 0.30 m glucose - Brainly.com
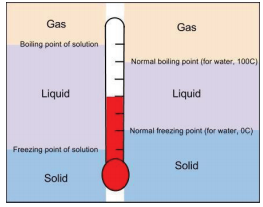
13.9: Freezing Point Depression and Boiling Point Elevation- Making Water Freeze Colder and Boil Hotter - Chemistry LibreTexts
Which of the following aqueous solutions has highest freezing point (assuming m=M)? (1)0.1M KNO3 (2)0.2M Na3PO4 (3)0.25M FeCl3 (4)0.01M Na2SO4