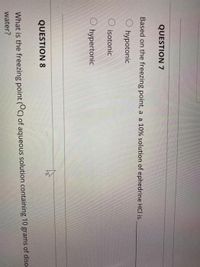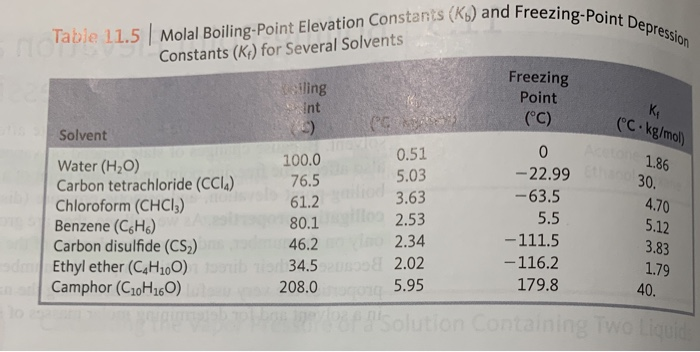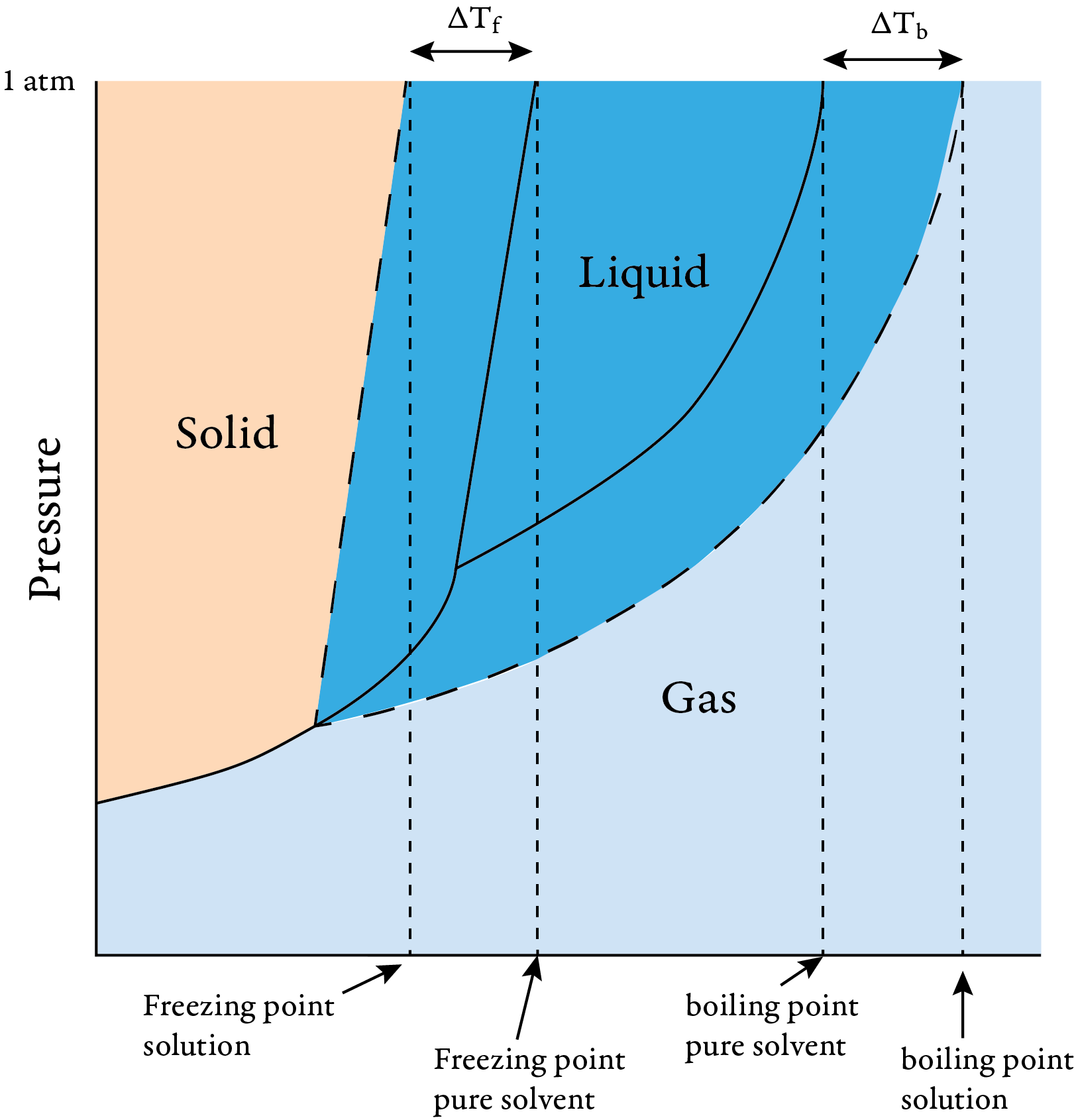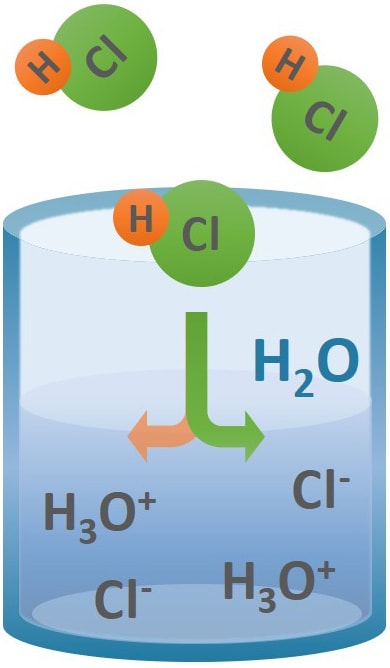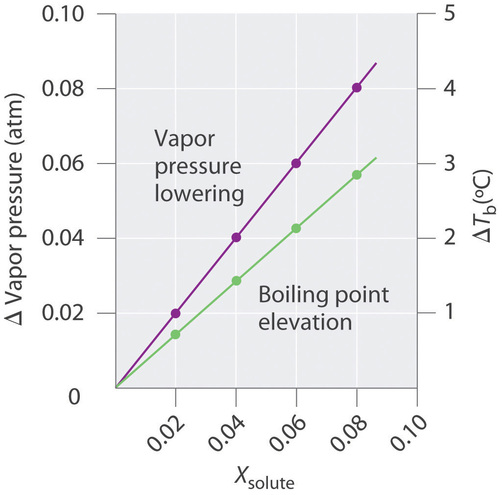
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts

SOLVED: Order the following aqueous solutions in ascending order of their expected freezing point. a) 0.050m CaCl2 b) 0.15m NaCl c) 0.10m HCl d) 0.050m HC2H3O2 e) 0.10 m C12H22O11
The depression in freezing point of 0.1 M aqueous solutions of HCl, CuSO4 and K2SO4 are in the ratio - Sarthaks eConnect | Largest Online Education Community
ACP - Trapping of HCl and oxidised organic trace gases in growing ice at temperatures relevant to cirrus clouds
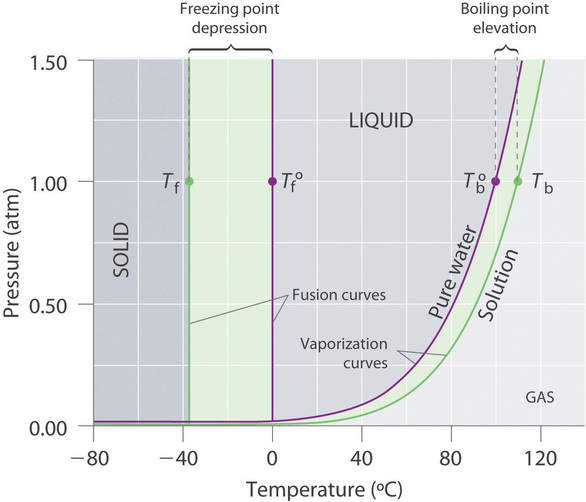
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts

11.51 | A 1.0 m solution of HCl in benzene has a freezing point of 0.4 °C. Is HCl an electrolyte in - YouTube

The depression in freezing point of 0.1 M aqueous solutions of HCl , CuSO4 and K2SO4 are in the ratio:

Calculate the depression in the freezing point of water when `10g` of `CH_(3)CH_(2)CHClCOOH` is ... - YouTube