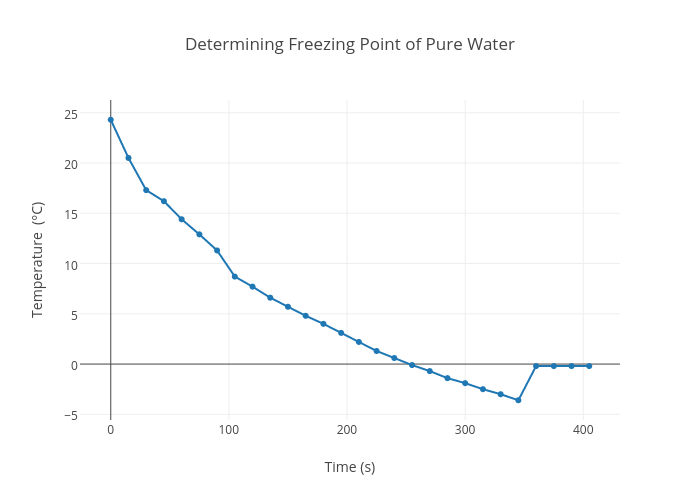
General Chemistry Online: FAQ: Solutions: How can freezing point depression be explained in terms of free energies?
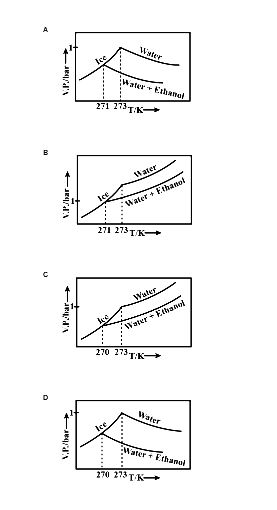
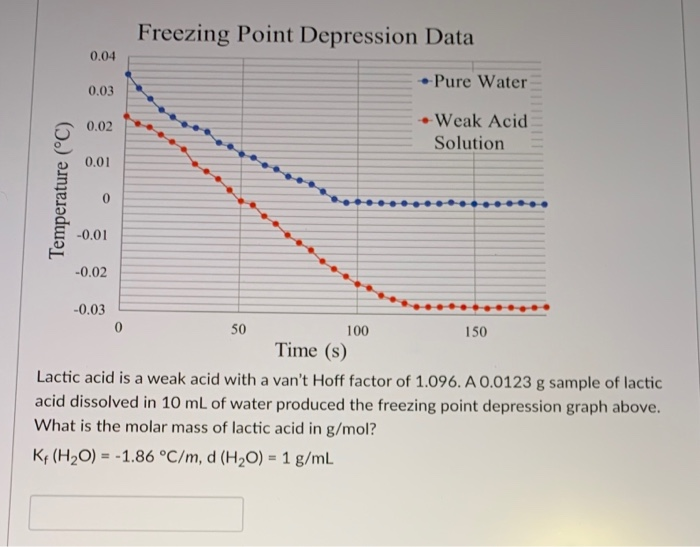
Pure water freezes at 273 K and 1 bar. The addition of 34.5 g of ethanol to 500g of water changes the freezing point of the solution. Use the freezing point depression

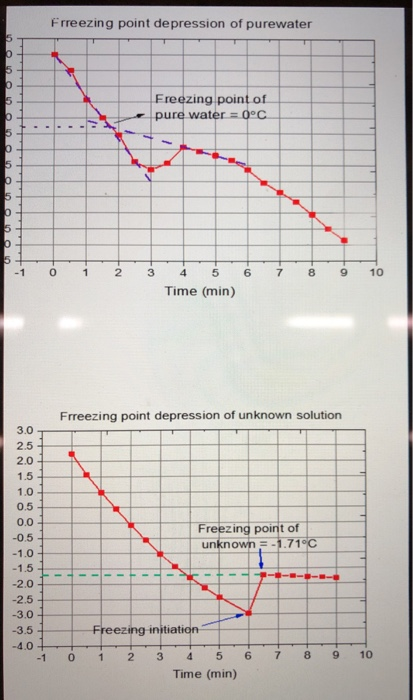
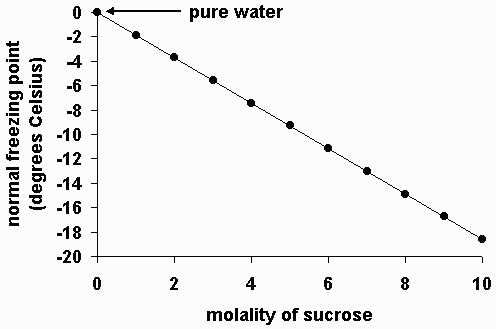
A 5% (by mass) of cane sugar in water has freezing point of 271K . Calculate the freezing point of 5% glucose in water if freezing point of water is 273.15K .

A 4% solution (w/w) of sucrose (M 342 g `mol^(-1)`) in water has a freezing point of 271.15K - YouTube

Pure water freezes at 273 K and 1 bar. The addition of 34.5 g of ethanol to 500 g of water changes the freezing point of the solution. Use the freezing point

A 5% (by mass) of cane sugar in water has freezing point of 271K . Calculate the freezing point of 5% glucose in water if freezing point of water is 273.15K .

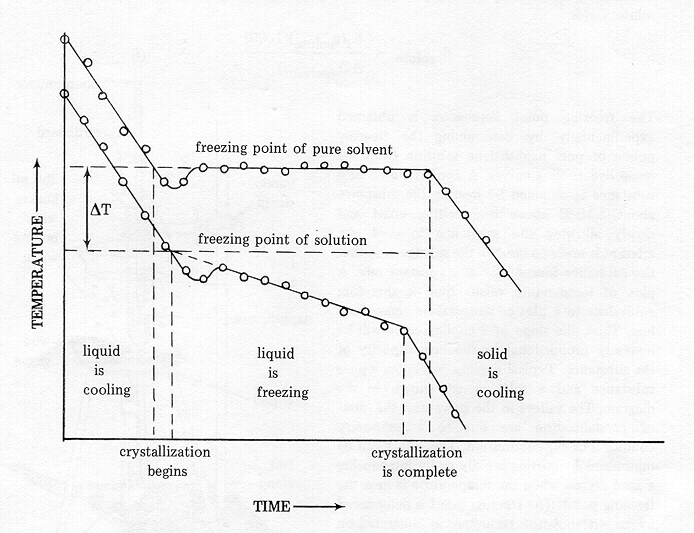
2 Typical freezing profile of frozen pure water and food systems. The... | Download Scientific Diagram
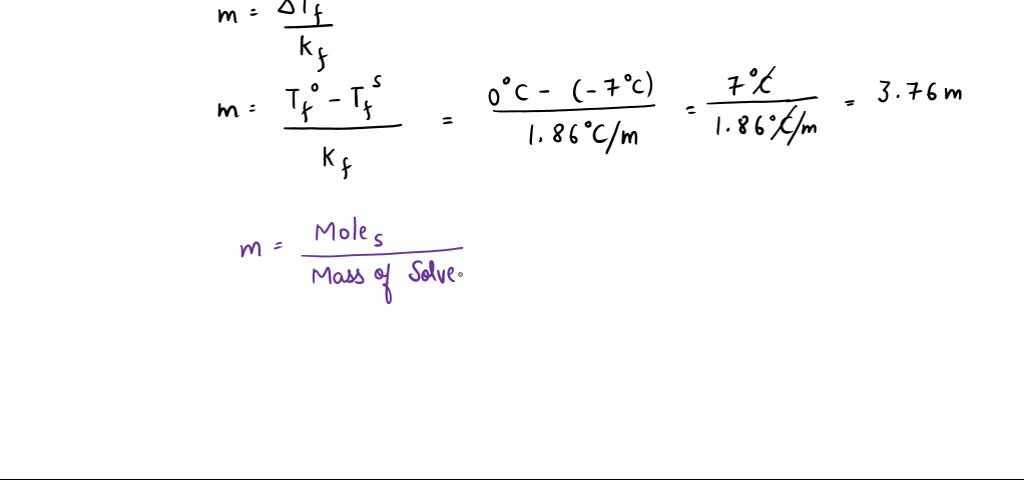
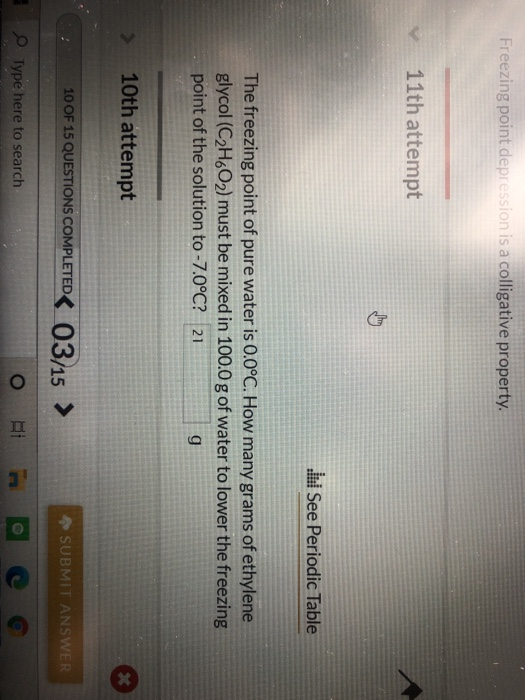
SOLVED: The freezing point of pure water is 0.0°C. How many grams of ethylene glycol (C2H6O2) must be mixed in 100.0 g of water to lower the freezing point of the solution

Comparison of pure water freezing using filtered MilliQ produced water... | Download Scientific Diagram
Science for beginners . ated solution of salt and water is —7°F., or 39° belowthe freezing point of pure water (Art. 16). If a mixtureof one-third salt and two-thirds crushed ice is

:max_bytes(150000):strip_icc()/the-freezing-point-of-water-609418_FINAL-01f50f5f4f7d4a39854bebcc59df1aa4.gif)