How to calculate the freezing point of an equimolar ethanol: water mixture using colligative properties - ECHEMI

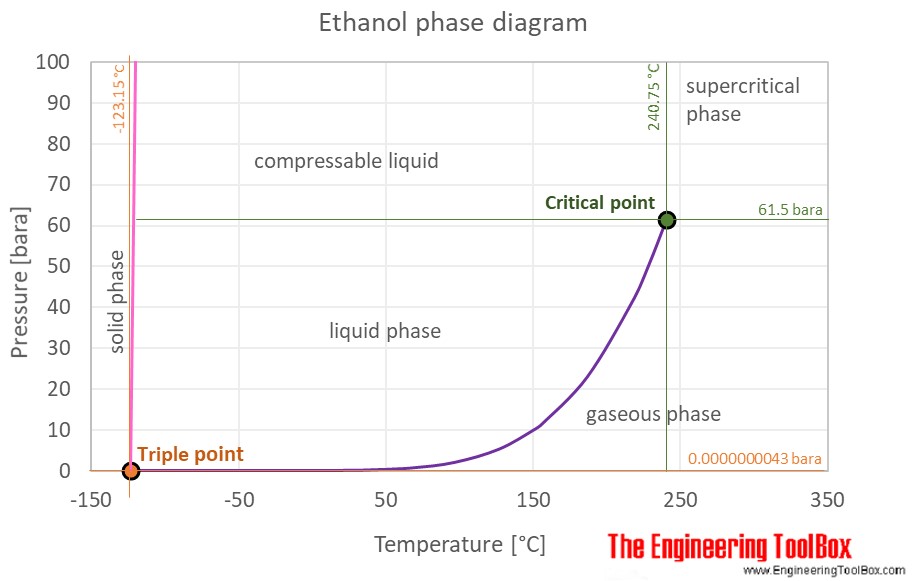
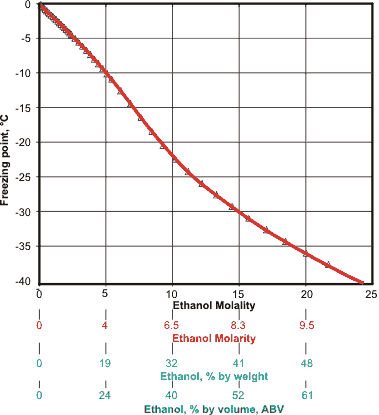
Freezing point diagram of an ethyl alcohol/water mixture (values from... | Download Scientific Diagram
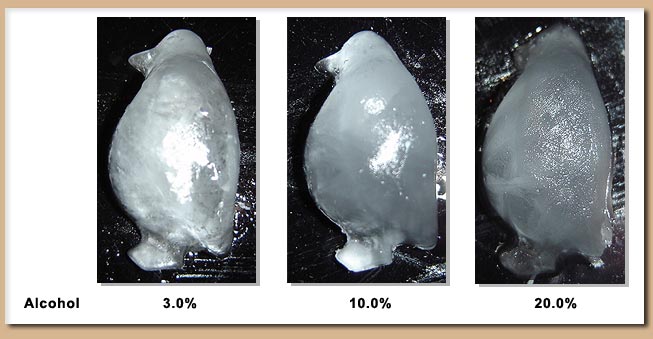
Scientific AmeriKen: Alcohol and Freezing - determining the freezing ability of water as a function of impurity!
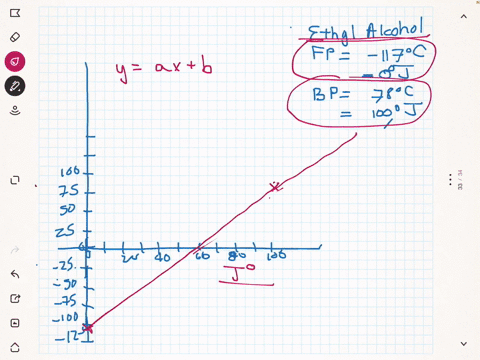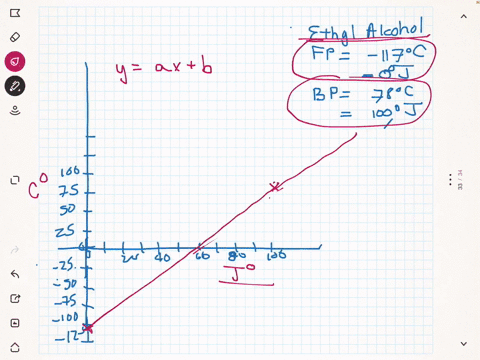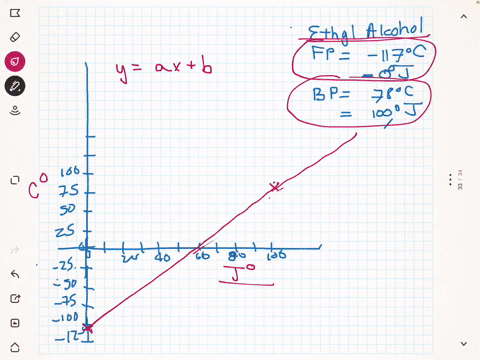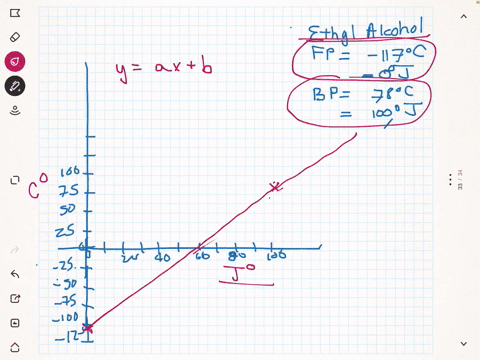
SOLVED:A different civilization on a distant planet has developed a new temperature scale based on ethyl alcohol. The freezing point of ethyl alcohol (-117^∘ C) is designated as 0^∘ J, and its
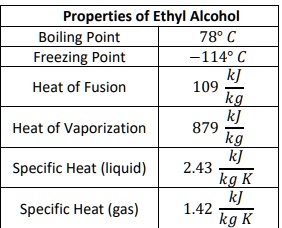
SOLVED: Properties of Ethyl Alcohol Boiling Point 78" Freezing Point 1140 Heat of Fusion 109 Heat of Vaporization 879 Specific Heat (liquid) 2.43 Specific Heat (gas) 1.42 kg K

thermodynamics - What mixing ratio of ethanol and acetone has the lowest freezing point? - Chemistry Stack Exchange

Freezing Point Depression: when 15.0 grams of ethyl alcohol (C(2)H(5)OH) is dissolved in 750 grams of formic acid, the freezing point of the solution is 7.20^(@)C. The freezing point of pure formic
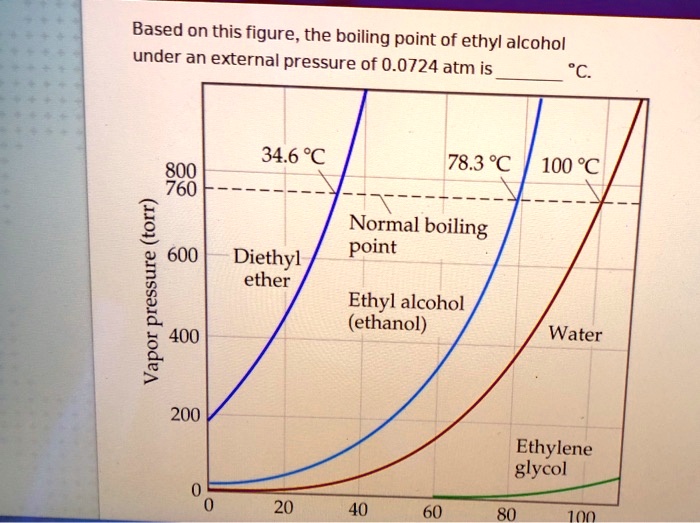
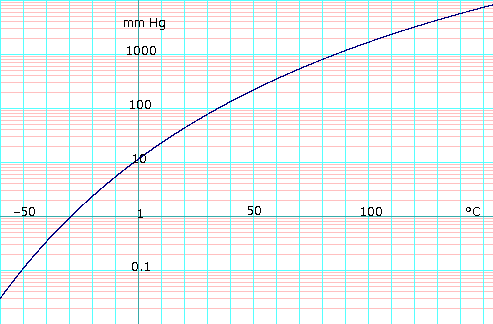
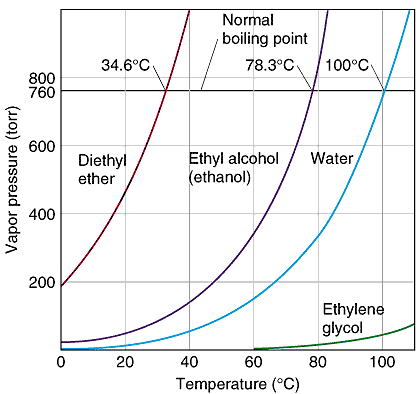
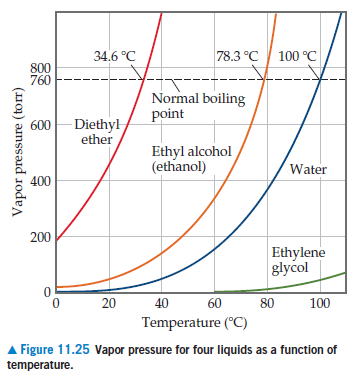
SOLVED: Based on this figure, the boiling point of ethyl alcohol under an external pressure of 0.0724 atm is 'C 34.6 *C 800 78.3 'C 100 % 760 0 Normal boiling 600

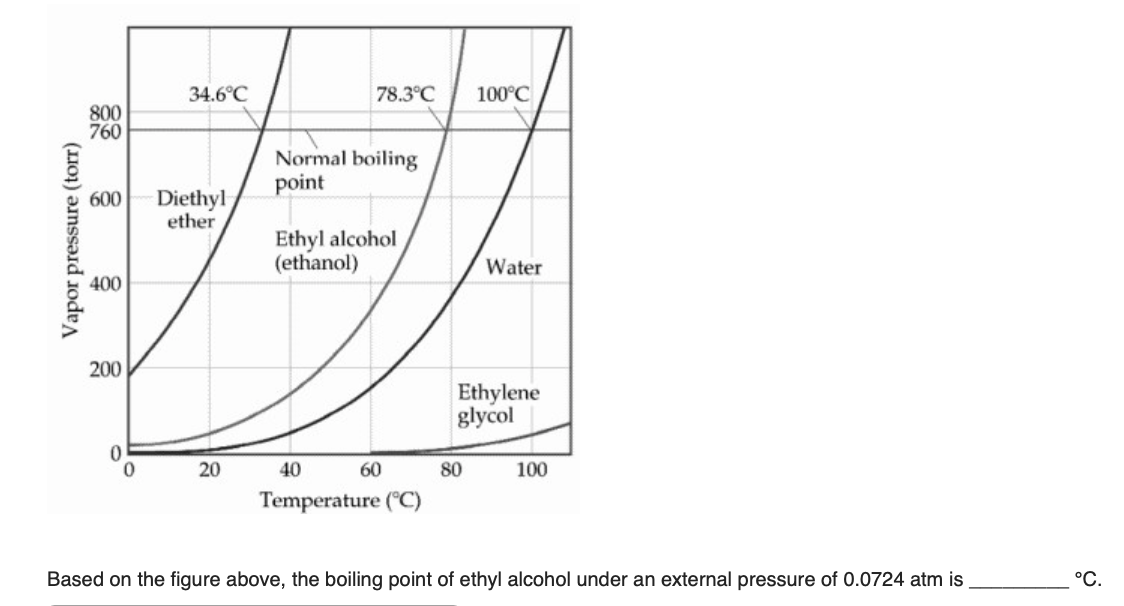

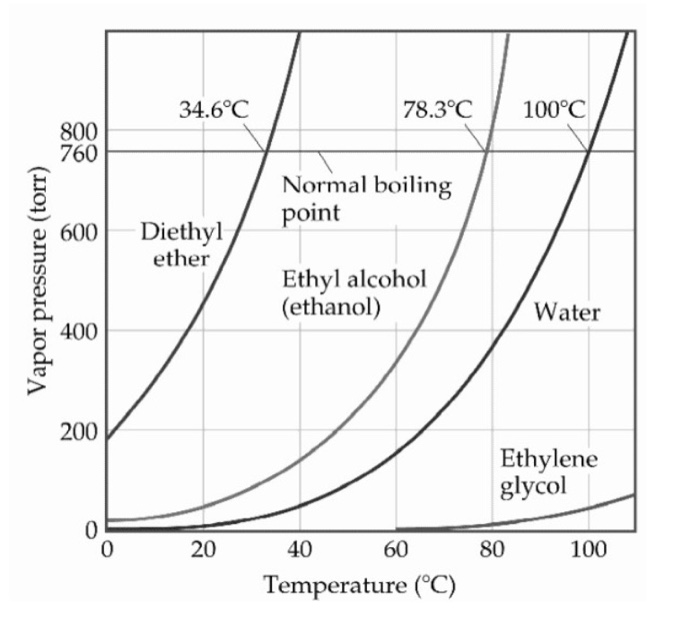
:max_bytes(150000):strip_icc()/GettyImages-1150475927-bc0edc49670e4adba0e80a4fc3320d8f.jpg)
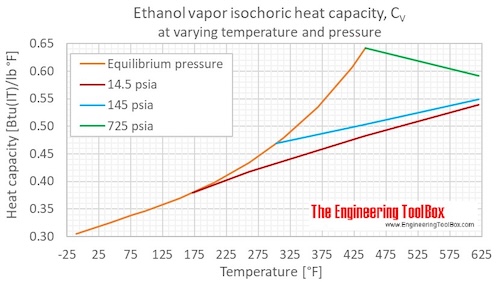



:max_bytes(150000):strip_icc()/will-liquor-freeze-760302_V3-1909ef0532f34ff0a2acee1a01b093c5.png)