All the COVID-causing virus in the world would fit in a coke can: Mathematician | Al Arabiya English

How to Make a Self-Freezing Coca-Cola Slushy (Or Any Kind of Instant Soda Slurpee) « Food Hacks :: WonderHowTo

How to Make a Self Freezing Coca-Cola (Or Any Instant-Soda Slurpee) : 6 Steps (with Pictures) - Instructables

How to Make a Self-Freezing Coca-Cola Slushy (Or Any Kind of Instant Soda Slurpee) « Food Hacks :: WonderHowTo

The Freezing Point Of Lysterin Mouthwash: Exploring Its Properties And Potential Benefits | DentalEHub.com
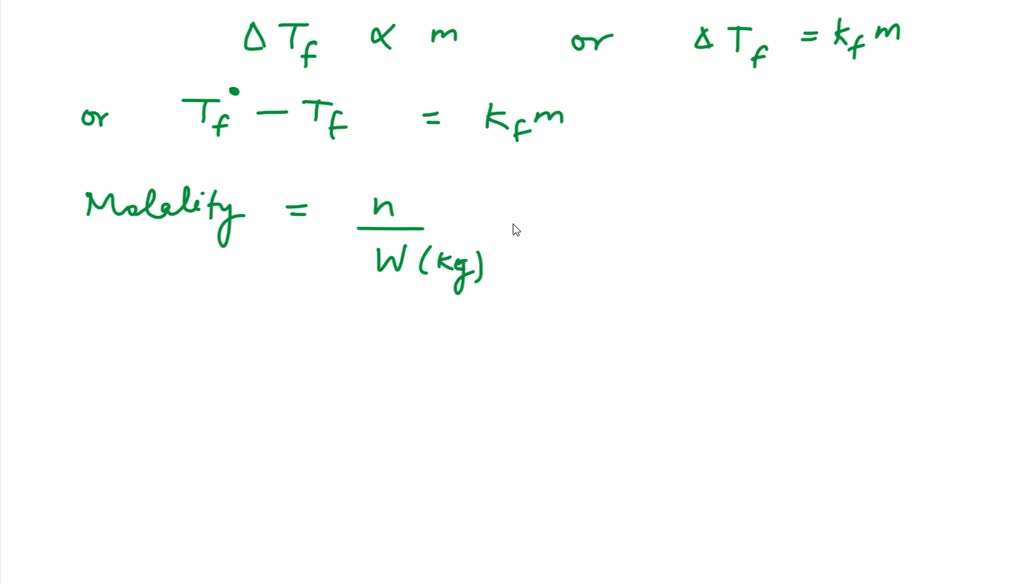
SOLVED: Coca-cola has 3.38 g sucrose (C12H22O11) in every 29.6 mL water (Kf = 1.86 °C/m). Calculate the freezing point of Coke if you let it go flat and assume that nothing

Temperature and Heat. The temperature of boiling water is 100º on the Celsius scale and 212º on the Fahrenheit scale. Look at the following temperatures. - ppt download