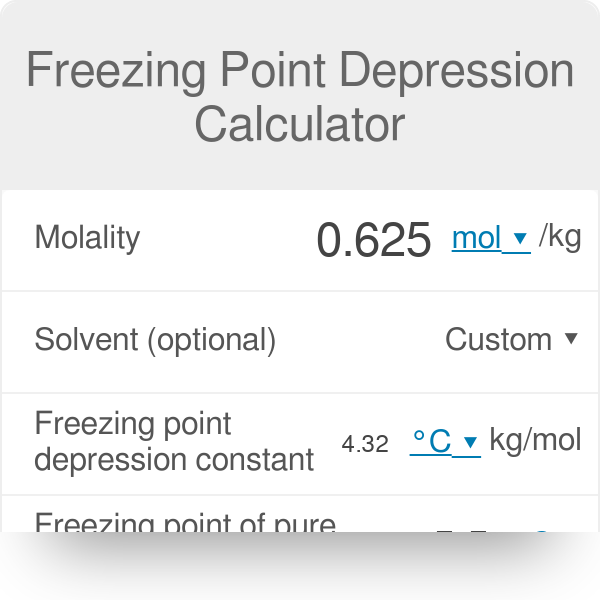
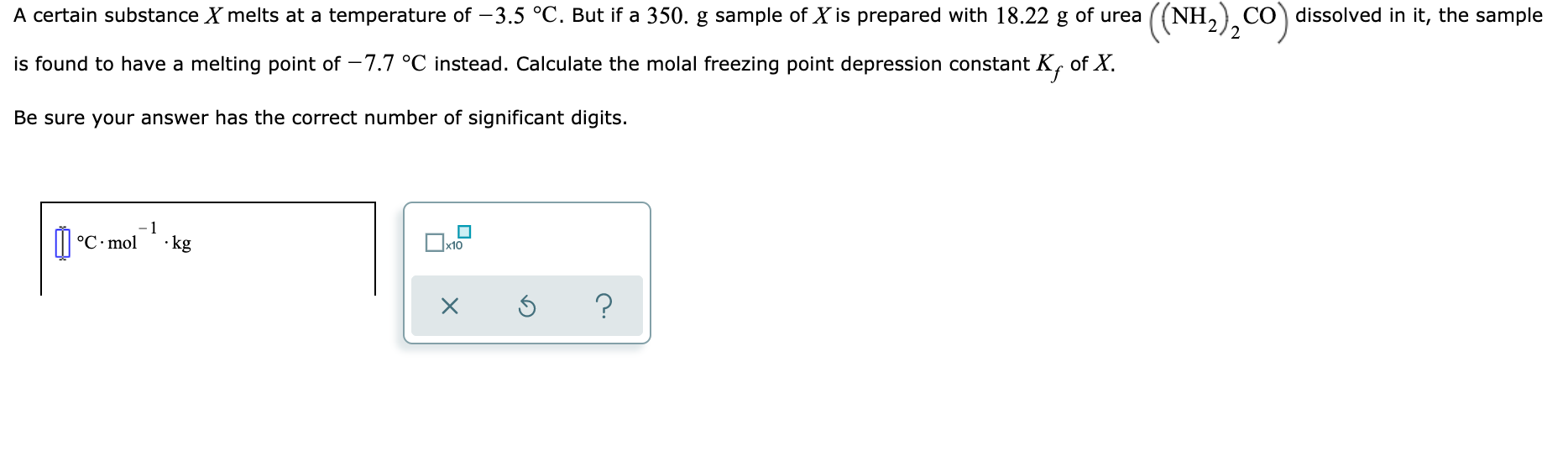
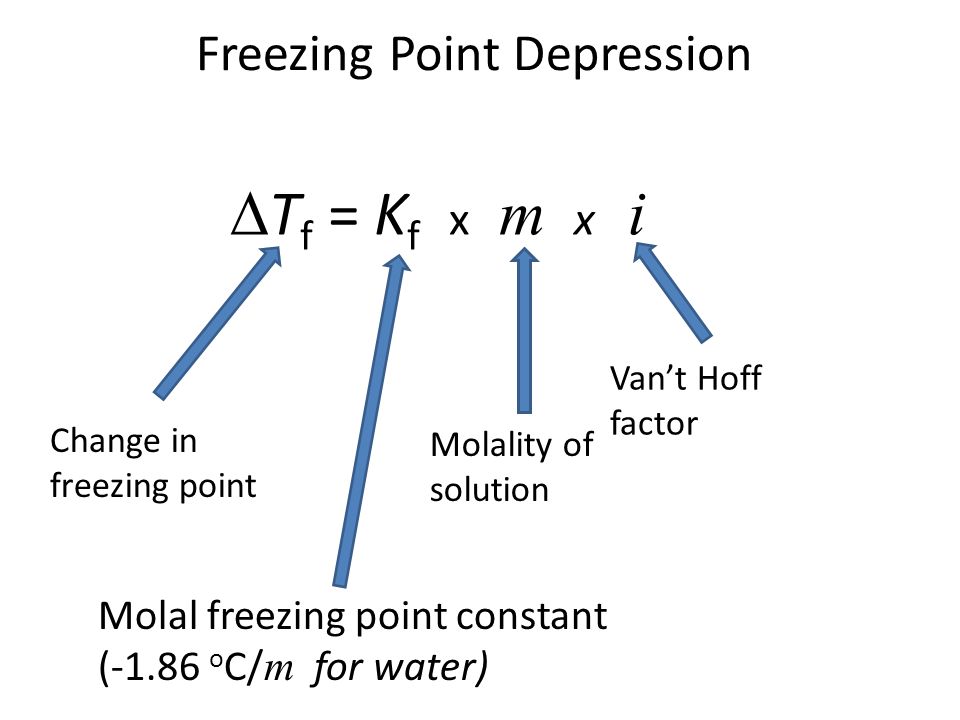
A brief introduction to freezing point depression THE COLLIGATIVE PROPERTIES OF MOLALITY AND FREEZING POINT DEPRESSION grownextgen.org. - ppt download

Calculate the freezing point and the boiling point at 1 atmosphere of a solution containing 30 g cane sugar (molecular mass 342 ) and 150 g water.Given : Kb = 0.513 and Kf = 1.86

45 g of ethylene glycol (C2H6O2) is mixed with 600 g of water. The freezing point of the solution is ( Kf for water is 1.86 K kg mol^-1 ).

Calculate the depression in the freezing point of water when `10g` of `CH_(3)CH_(2)CHClCOOH` is ... - YouTube

A 5% solution (by mass) of urea in water has freezing point of 271.52 K . Calculate the freezing point of 5% glucose in water if freezing point of pure water is 273.15K

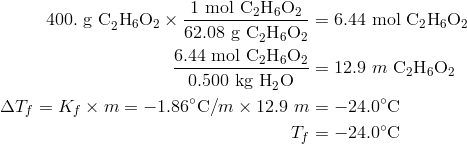
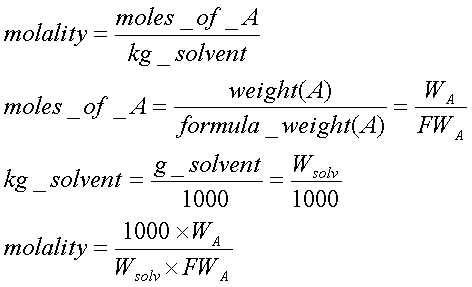


![Solved 8. [15 points] This problem concerns the colligative | Chegg.com Solved 8. [15 points] This problem concerns the colligative | Chegg.com](https://media.cheggcdn.com/media/86f/86fd907b-0a68-4171-b9c7-119b7fa8da27/phpO079OR.png)