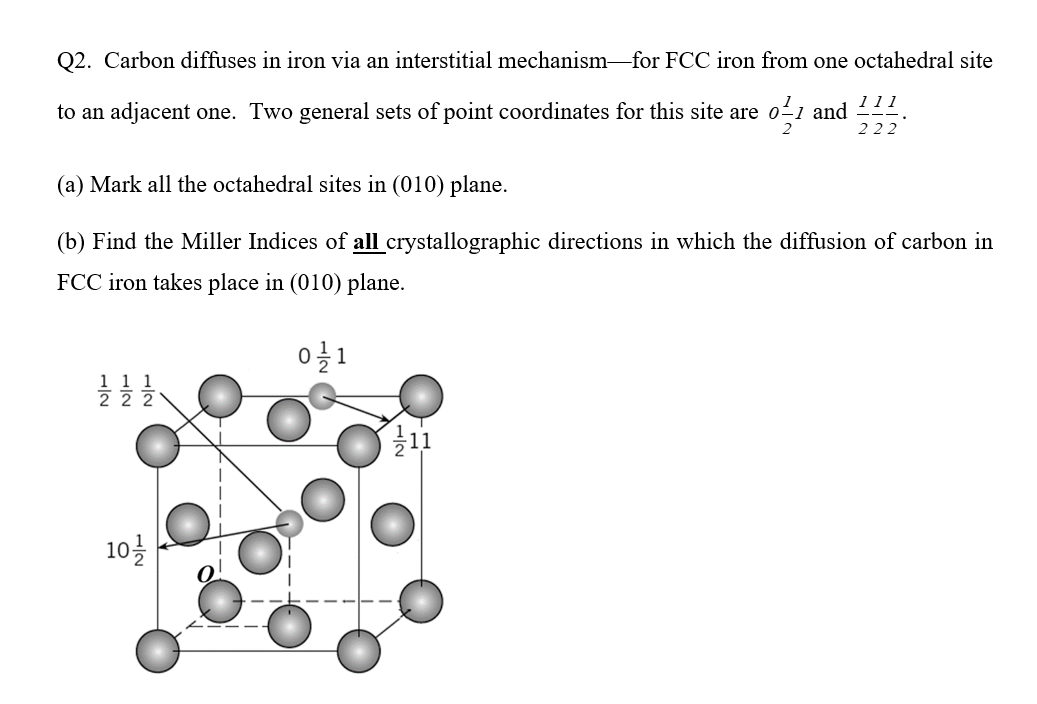
SOLVED: The lattice parameter (aFcc) 3.571 x 10/0 m for FCC iron; (aecc) 2.866 X 10-10 m for BCC iron; Radius of Iron atom (rre) 1.241 X 1010 mj Radius of Carbon atom (rc) 0.71 x 1010 mj
Point defect interactions in iron lattice: a first-principles study - RSC Advances (RSC Publishing) DOI:10.1039/C6RA05969D
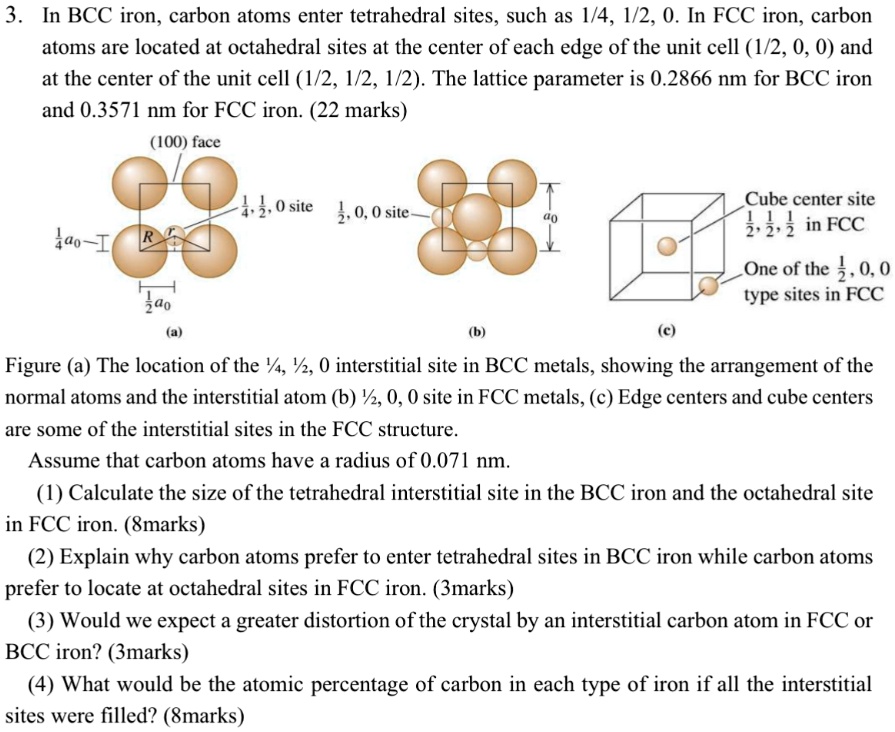
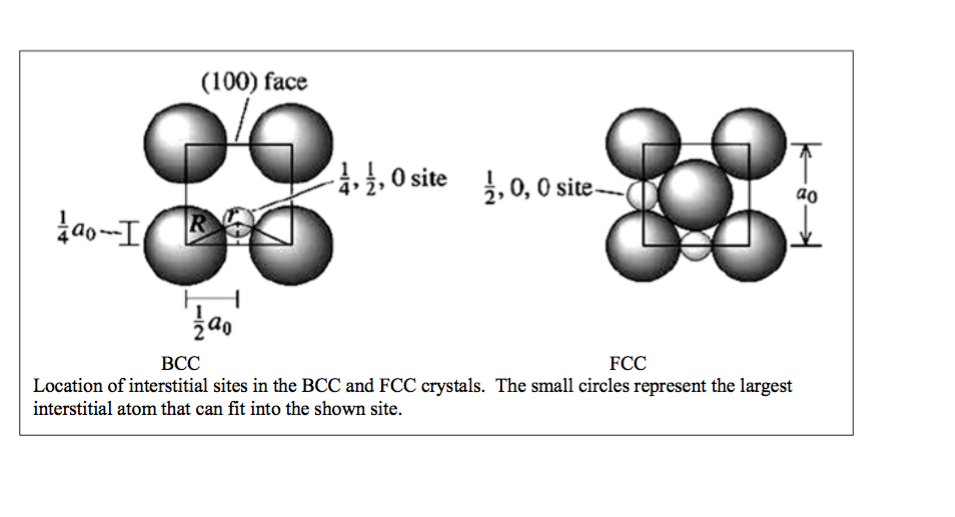
SOLVED: 3 In BCC iron, carbon atoms enter tetrahedral sites, such as 1/4, 1/2, 0. In FCC iron, carbon atoms are located at octahedral sites at the center of each edge of

Schematics of ͑ a ͒ fcc-Pd ͑ 110 ͒ , ͑ b ͒ bcc-Fe ͑ 110 ͒ , and ͑ c ͒... | Download Scientific Diagram
1: Crystallographic structure of Face centered cubic (FCC) iron and... | Download Scientific Diagram

Hw5 solutions - Professor Gopalan - Excerpts from this work may be reproduced by instructors for - Studocu
What Is the Difference Between FCC and BCC? (Crystal Structure, Properties, Interstitial Sites, and Examples) – Materials Science & Engineering

In FCC iron, carbon atoms are located at octahedral sites at the center of each edge of the unit cell (1/2,0,0) and at the center of the unit cell (1/2,1/2,1/2) . Illustrate
![PDF] CONCENTRATION-AND TEMPERATURE-DEPENDENT DIFFUSION COEFFICIENT OF CARBON IN FCC IRON MATHEMATICALLY DERIVED FROM LITERATURE DATA | Semantic Scholar PDF] CONCENTRATION-AND TEMPERATURE-DEPENDENT DIFFUSION COEFFICIENT OF CARBON IN FCC IRON MATHEMATICALLY DERIVED FROM LITERATURE DATA | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/36b506767f0e891a9614cc4fa61333895faa7e7e/3-Table1-1.png)
PDF] CONCENTRATION-AND TEMPERATURE-DEPENDENT DIFFUSION COEFFICIENT OF CARBON IN FCC IRON MATHEMATICALLY DERIVED FROM LITERATURE DATA | Semantic Scholar
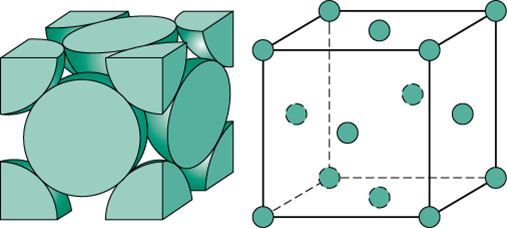
Iron crystallizes in a face-centered cubic system. If the radius of an iron atom is 1.26 A (angstroms), what is the edgelength of the unit cell? What is the density of iron

Volume Change During BCC to FCC Iron transformation//Calculation of Volume change in transformations - YouTube

First-principles study of interaction between vacancies and nitrogen atoms in fcc iron - ScienceDirect

Crystal structures of fcc, hcp, and dhcp iron hydrides: grey and blue... | Download Scientific Diagram

First-principle investigation of electronic structures and interactions of foreign interstitial atoms (C, N, B, O) and intrinsic point defects in body- and face-centered cubic iron lattice: A comparative analysis - ScienceDirect






![Crystallography of Iron Films [IAP/TU Wien] Crystallography of Iron Films [IAP/TU Wien]](https://www.iap.tuwien.ac.at/www/_media/surface/stm_gallery/fcc_bcc.gif)


![Solved] When BCC iron is heated, it changes to FCC iron resulting in Solved] When BCC iron is heated, it changes to FCC iron resulting in](https://storage.googleapis.com/tb-img/production/21/10/F1_Jaideep_Ravi_19.10.21_D8.png)