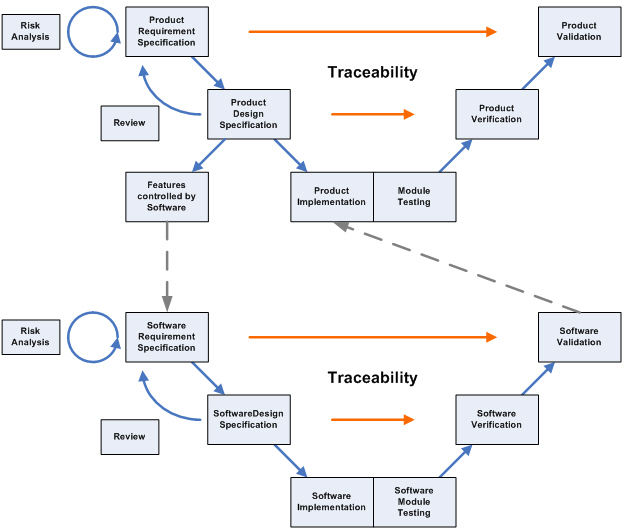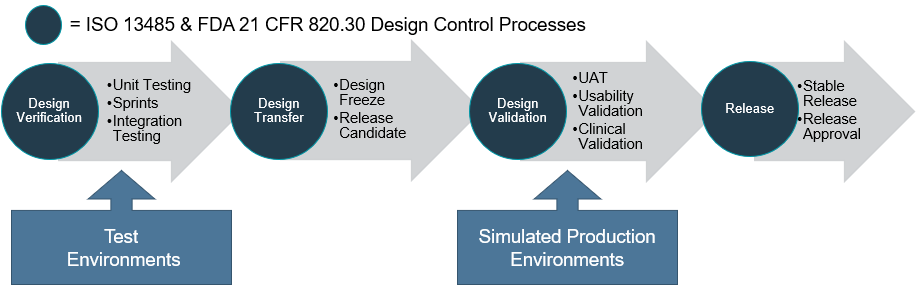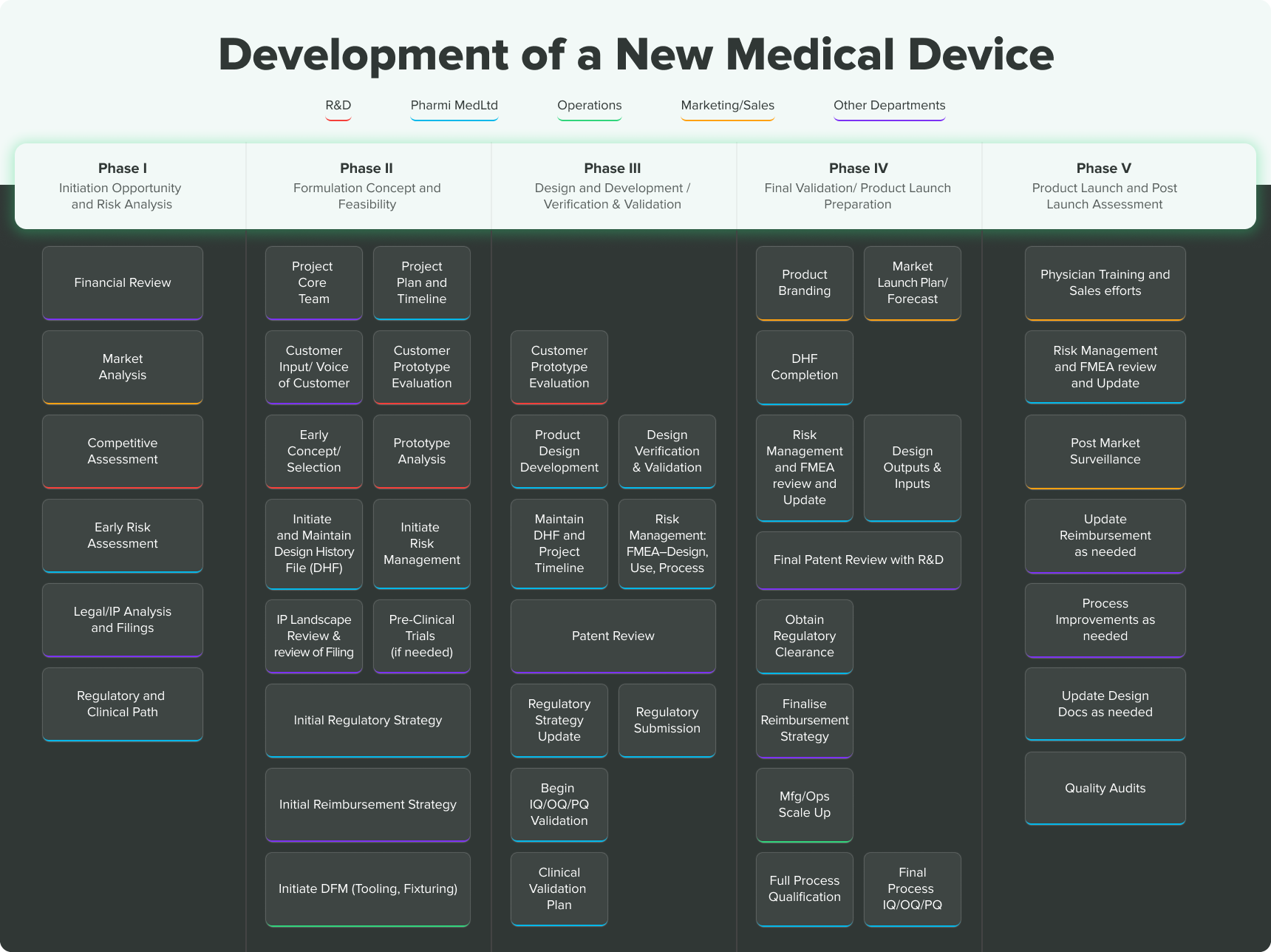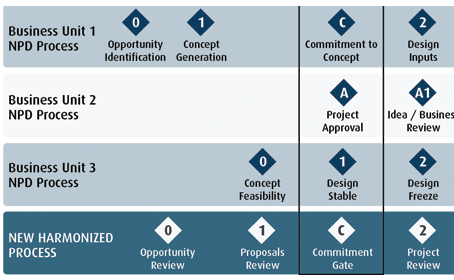
Usability Engineering: key to ensuring your medical device is ready for clinical evaluations - eg technology

Azzur Group - One of the areas we serve in #lifesciences is the Medical Devices and Diagnostics industry. By balancing project management and design controls, Azzur Group helps start, scale, and sustain