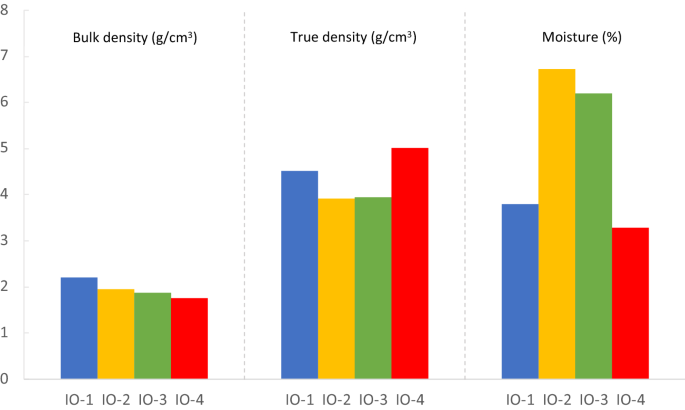
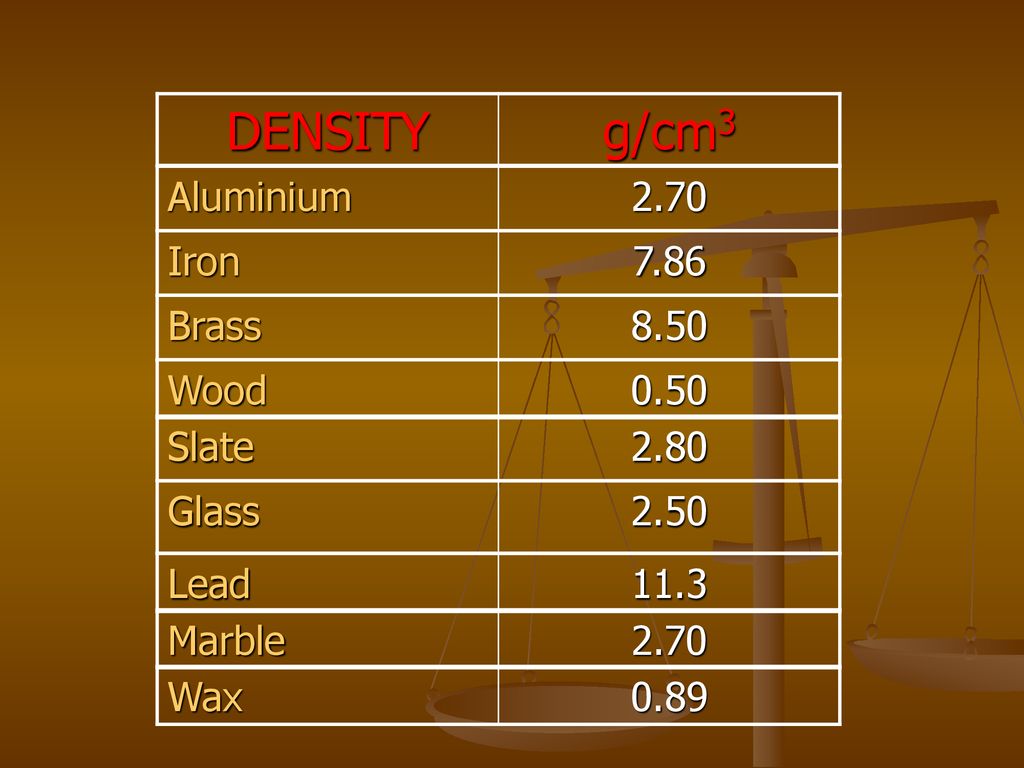
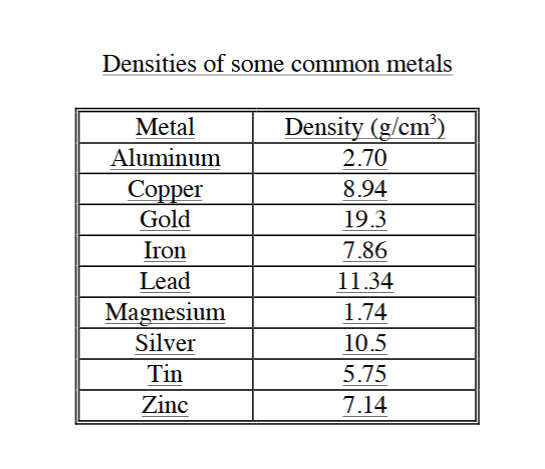
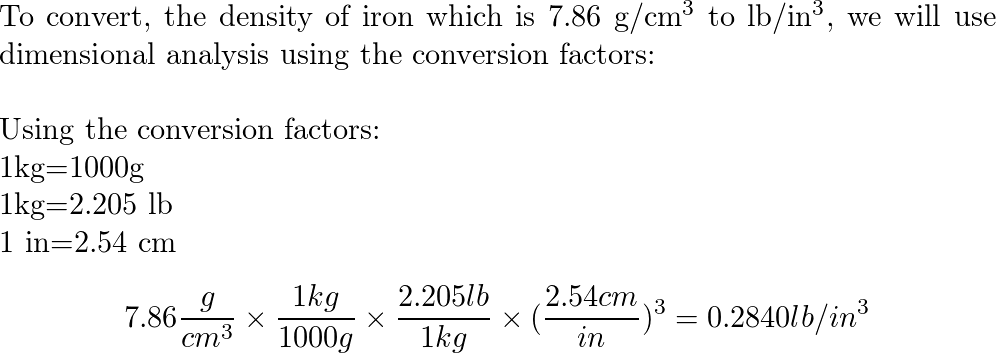The density of iron is 7.87 g/mL. What is the mass of a 3.00 cm × 4.00 cm × 1.00 cm block of iron? - Brainly.in
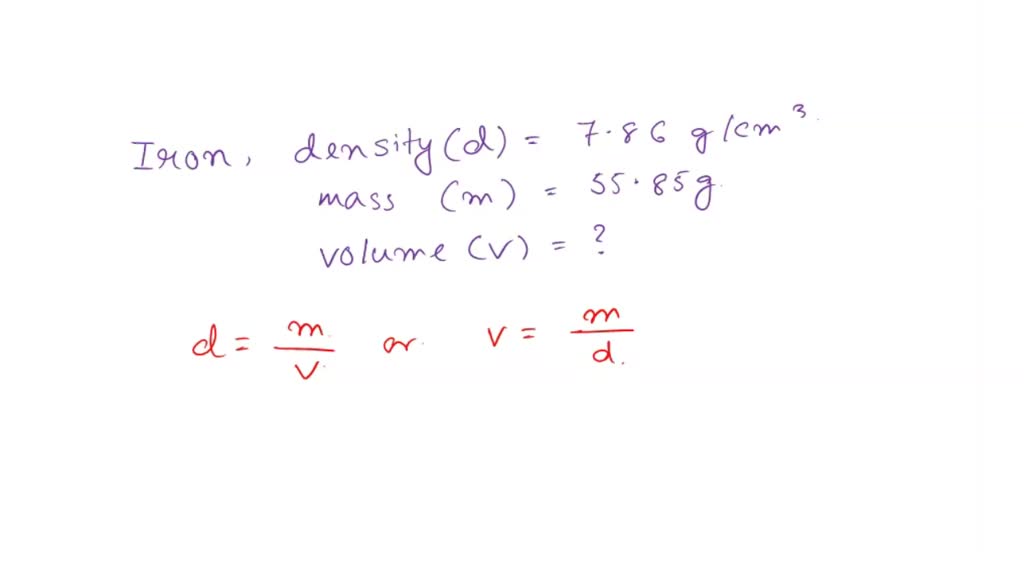
SOLVED: Iron has a density of 7.86 g/cm3. The volume occupied by 55.85 g of iron is Group of answer choices 2.8 cm3 439 cm3 0.141 cm3 7.11 cm3
Table of Densities Material Density g/cm3 Water 1.0 Alcohol 0.8 Diamond 3.5 Iron 7.9 Gold 18.9 Lead 11.3 English oak 0.7 Ice 0.9
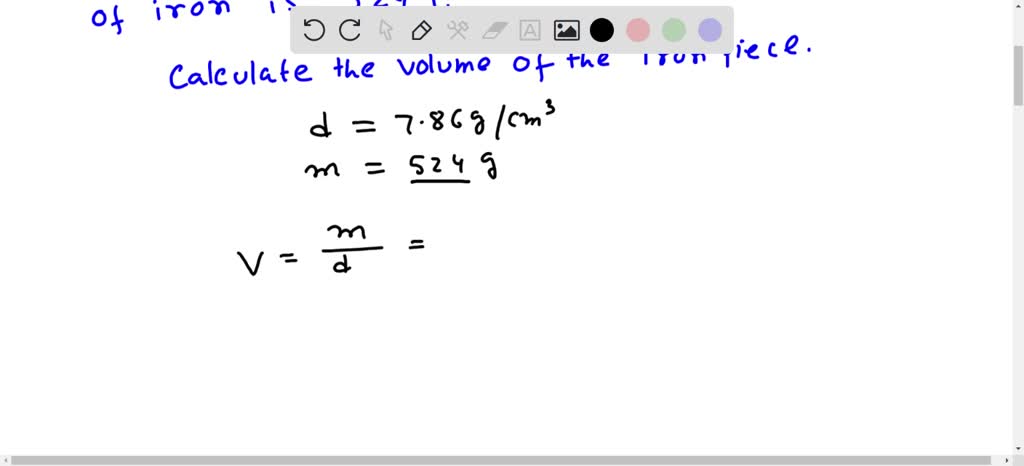
SOLVED: The density of iron is 7.86 g/cm3. What is the volume in milliliters (mL) of an irregularly shaped piece of iron that has a mass of 524 g?
The density of iron crystal is 8.54 gram cm^–3. If the edge length of unit cell is 2.8A° and atomic mass is 56 gram mol^–1. - Sarthaks eConnect | Largest Online Education Community

Iron has a body centred cubic unit cell with the cell dimension of 286.65 pm. Density of iron is 7.87 g cm ^-3 . Use this information to calculate Avogadro's number?(Atomic mass

Magnetic moment of an iron atom is 1.75 × 10^-23 A - m^2. If dipole moments of all the atoms of an iron bar of 6cm × 1 cm × 1 cm

If the density of iron is 7.85 g cm^(-3) and density of water is 1000 kg m-3, then the relative density of - Brainly.in

If the density of iron is 7.85 g cm^(-3) and density of water is 1000 kg m-3, then the relative density of iron is .