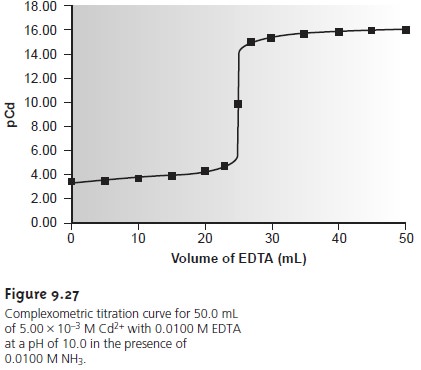
Question 17. Define EDTA titration and types of complexometric titration. by S P Kushwaha, India. - YouTube
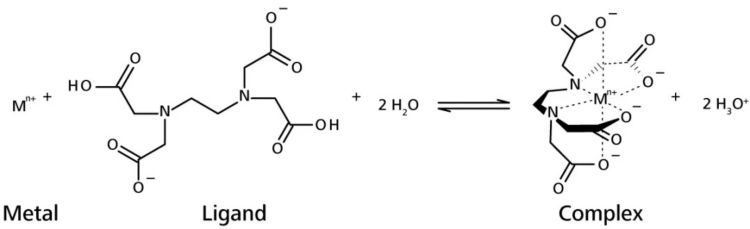
Complexometric titrations: new reagents and concepts to overcome old limitations - Analyst (RSC Publishing)

OneClass: I just need help with part B and how to calculate it. Using EDTA to titrate metal ions ...

Compleximetry.ppt - COMPLEXOMETRIC TITRATIONS General introduction EDTA titrations Advantages and limitations of EDTA as the titrant Absolute and | Course Hero

SOLVED: 3 Back Titration: Ni?+ was determined in a complexometric titration using EDTA at pH 5.5 and xylenol orange as the indicator: The unknown solution containing 25.00 mL of Ni2+ was first