
Nonclinical Information in the Common Technical Document: Opportunities for Content Reuse Peggy Zorn, MPI Research Susan Mattano, Pfizer, Inc. - ppt download

SUB04: Preparing Submissions in the Common Technical Document (CTD) Format | Zenosis – Learning for Life




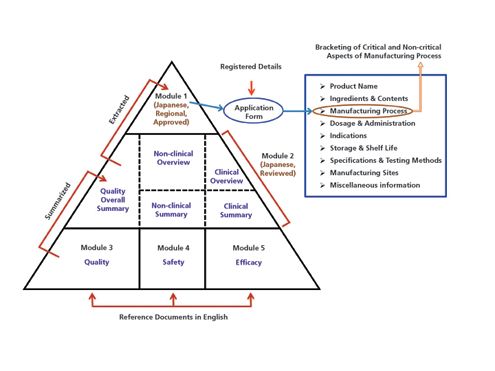

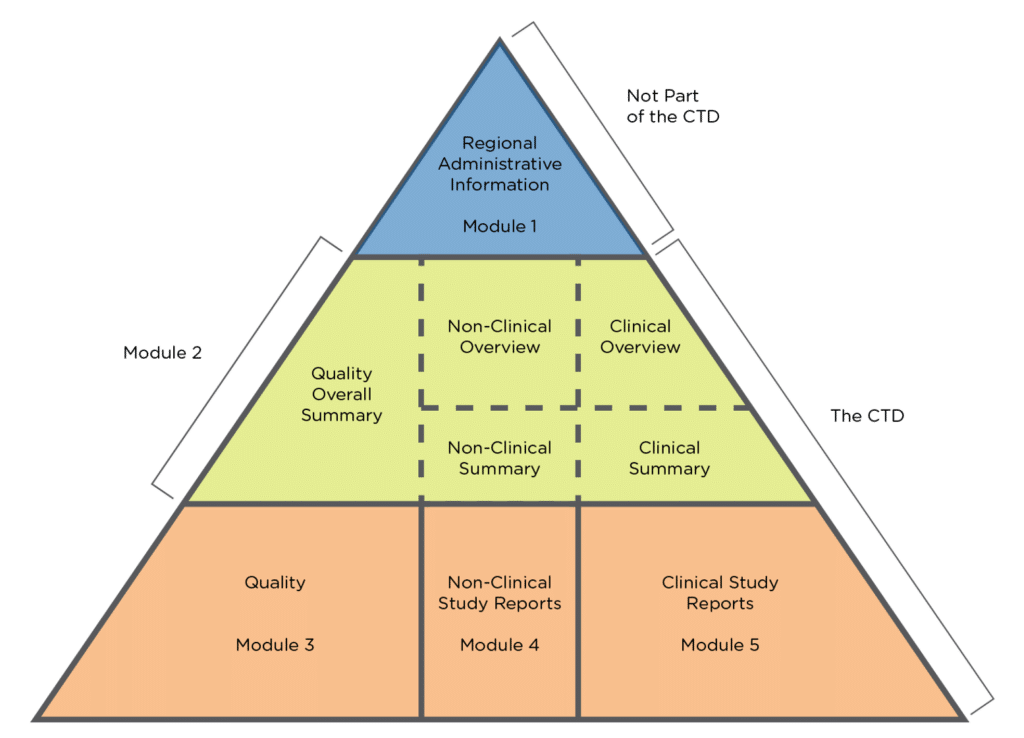

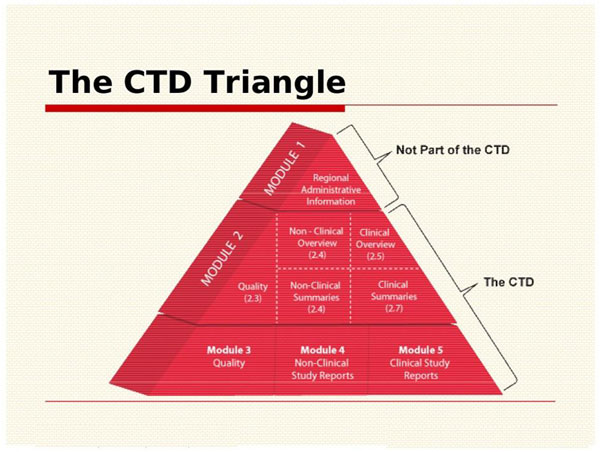
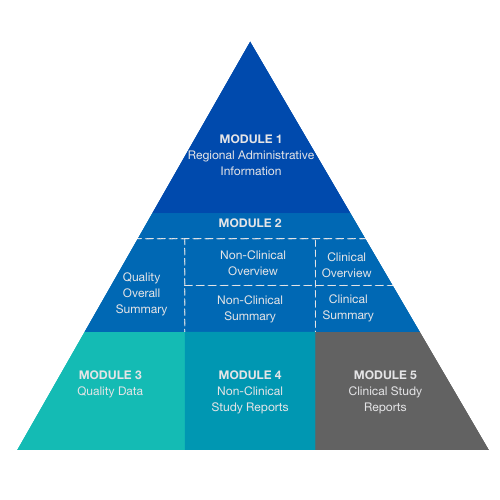
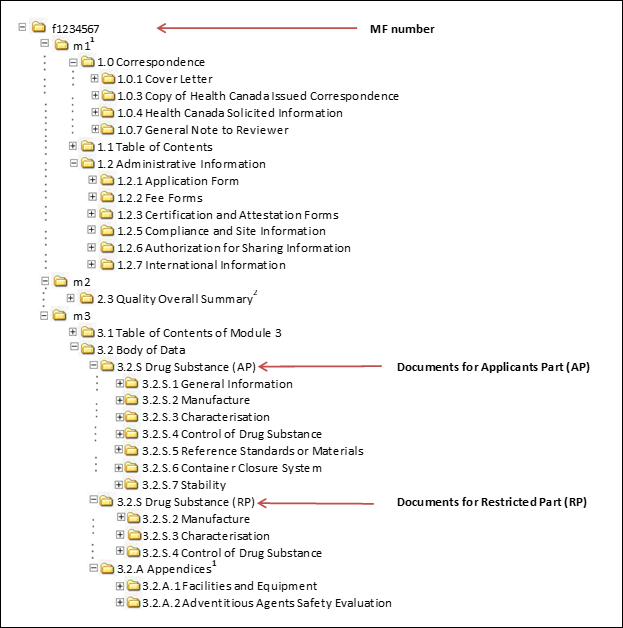


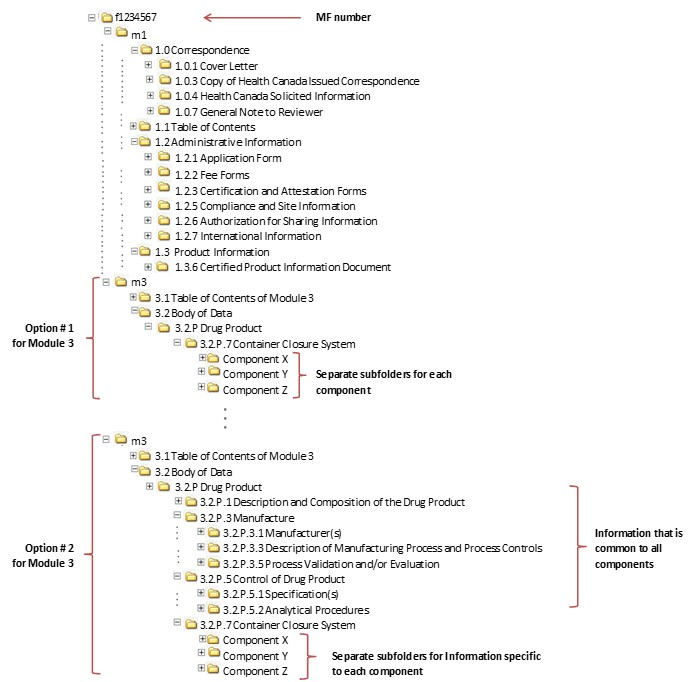
![Common technical document triangle [7] | Download Scientific Diagram Common technical document triangle [7] | Download Scientific Diagram](https://www.researchgate.net/publication/320372658/figure/fig1/AS:550839028203520@1508341666076/Common-technical-document-triangle-7.png)


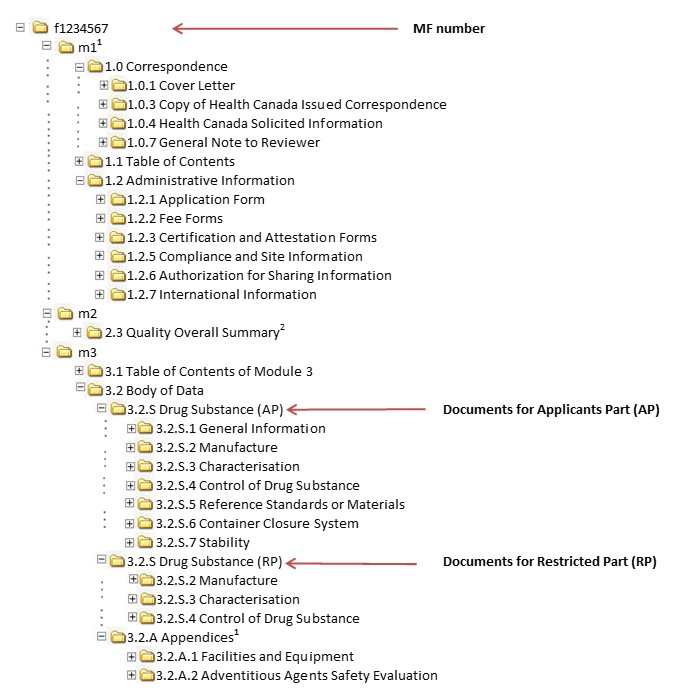
(1).png)

