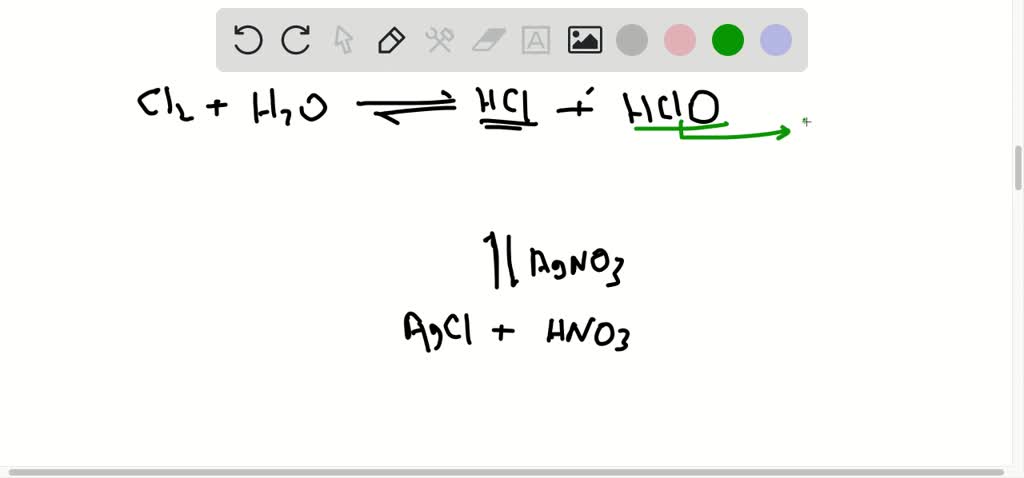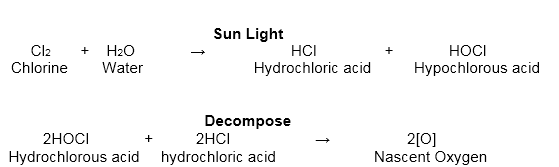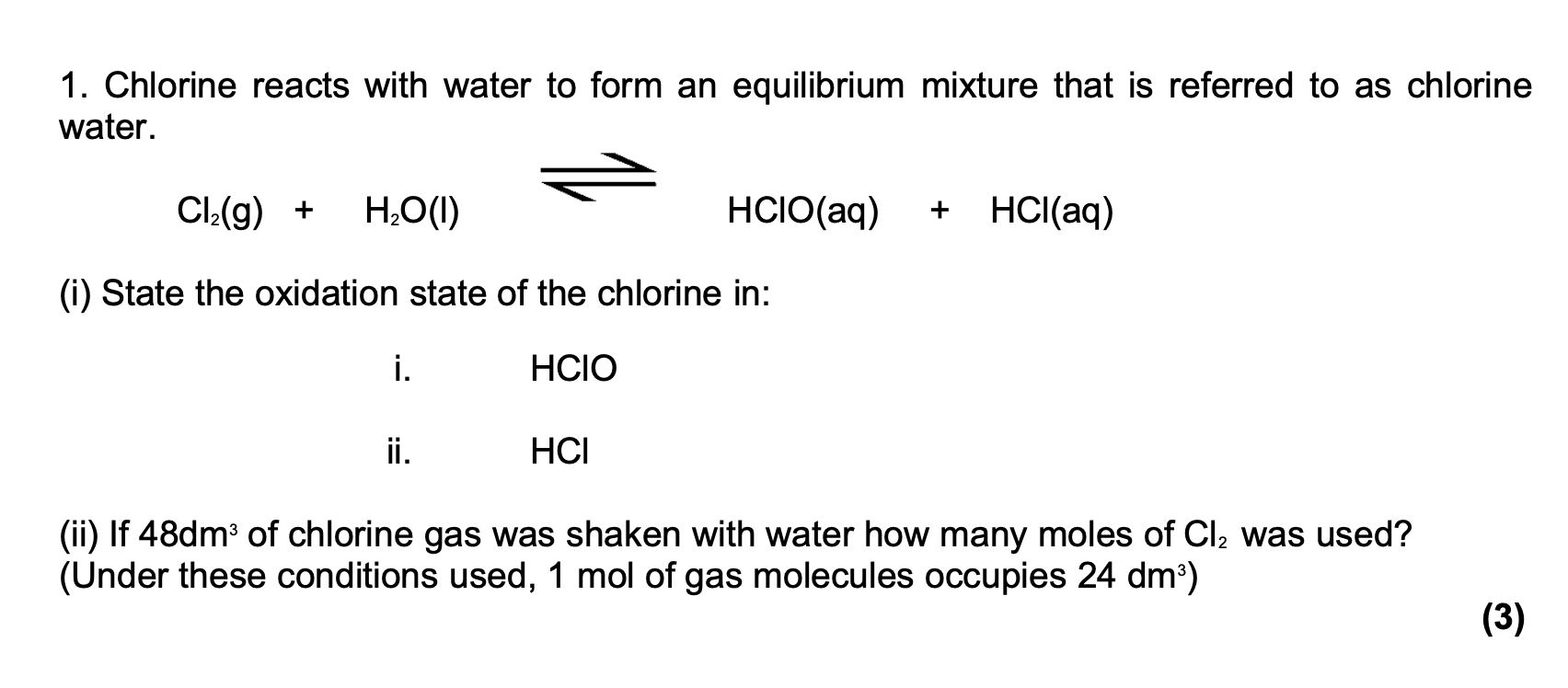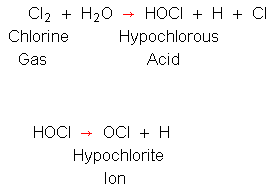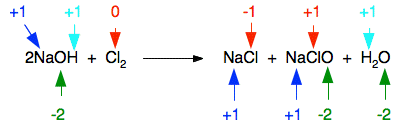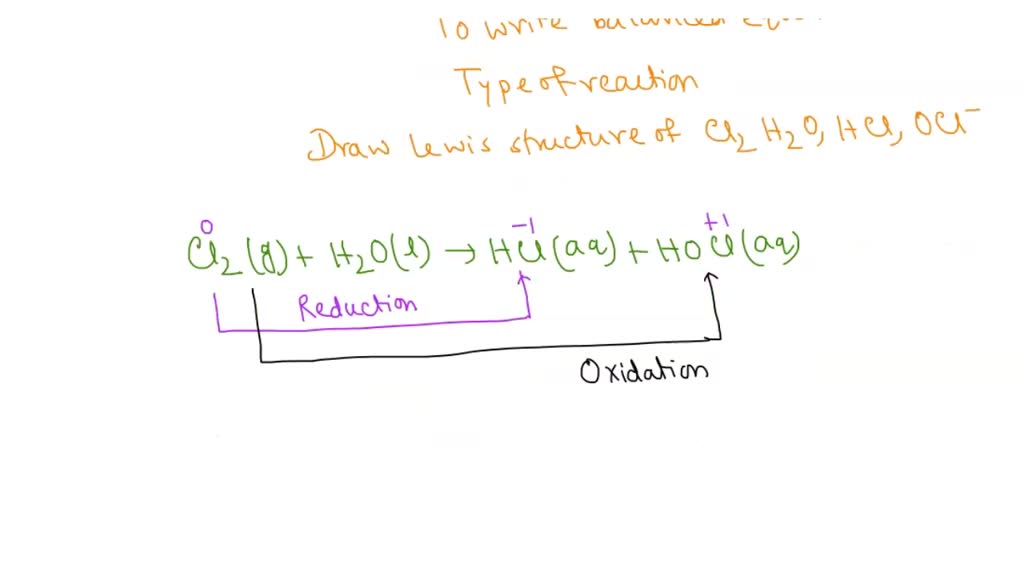
SOLVED: The reaction of chlorine gas with water results in the formation of aqueous hydrochloric acid and aqueous hypochlorous acid. Write the balanced chemical equation for the reaction of chlorine gas with
When sodium chloride dissolve in water, sodium chloride dissociates into Sodium and chlorine then why sodium does not react with water to form sodium hydroxide?

Reactions of chlorine with inorganic and organic compounds during water treatment—Kinetics and mechanisms: A critical review - ScienceDirect

What is cause of bleaching action of chlorine water ? Explain it with chemical equation. - Brainly.in