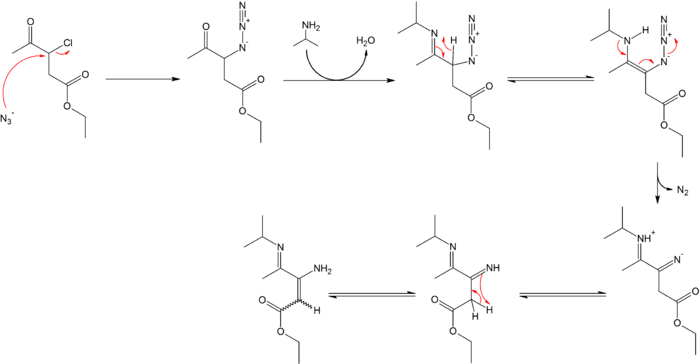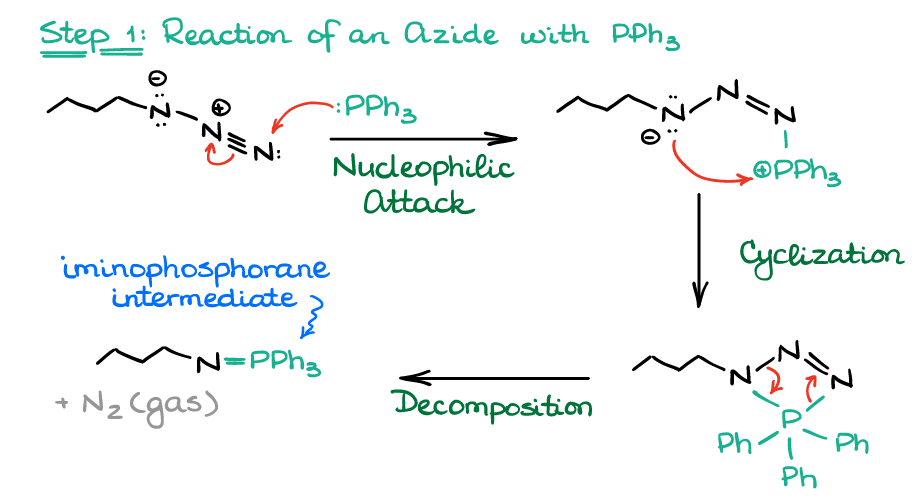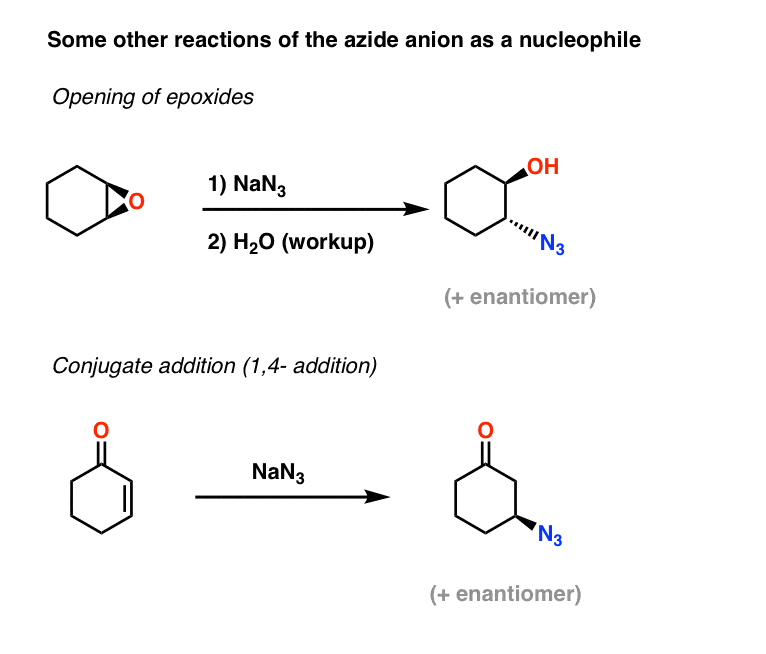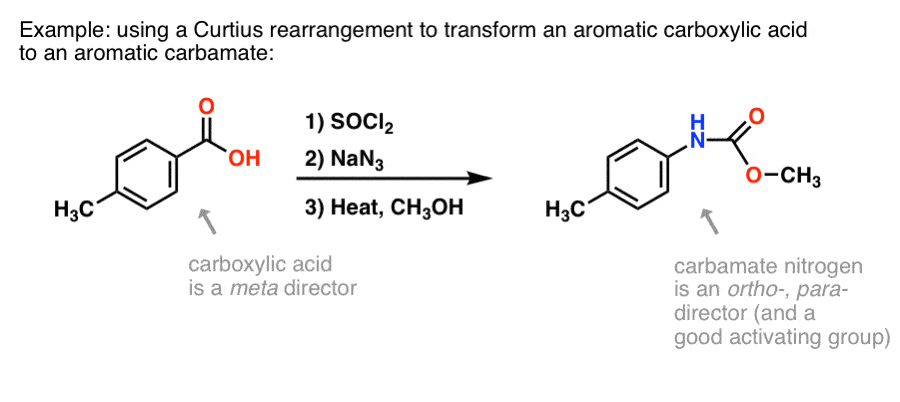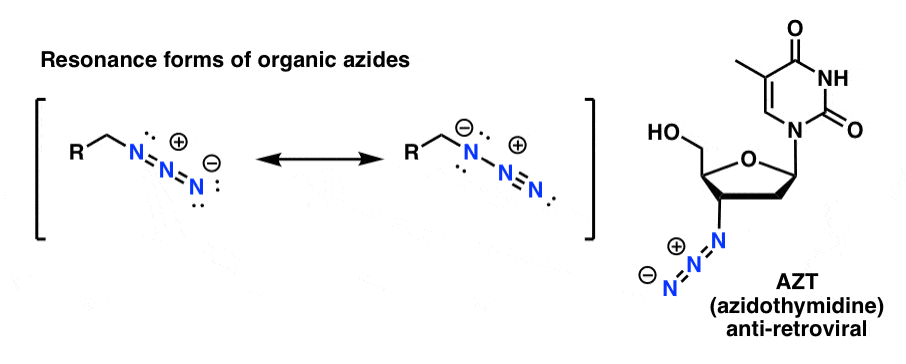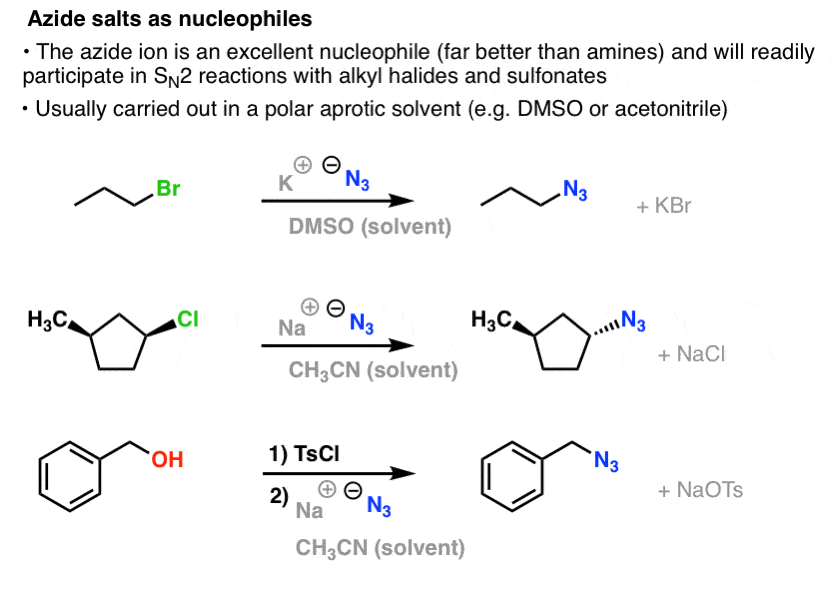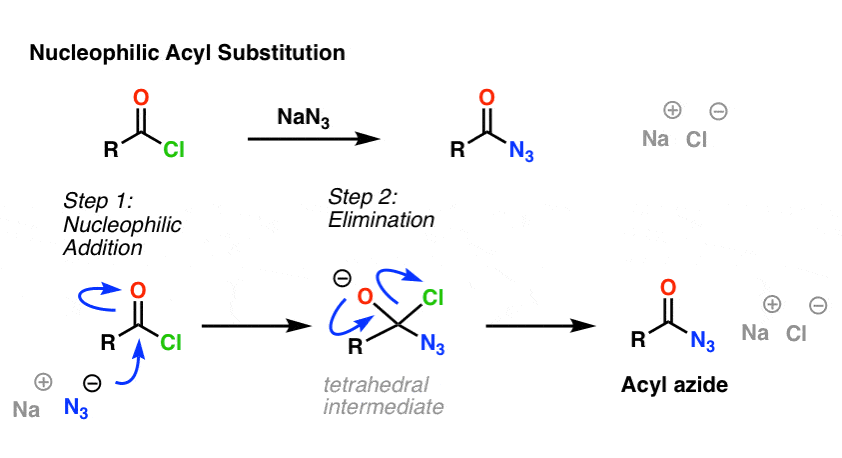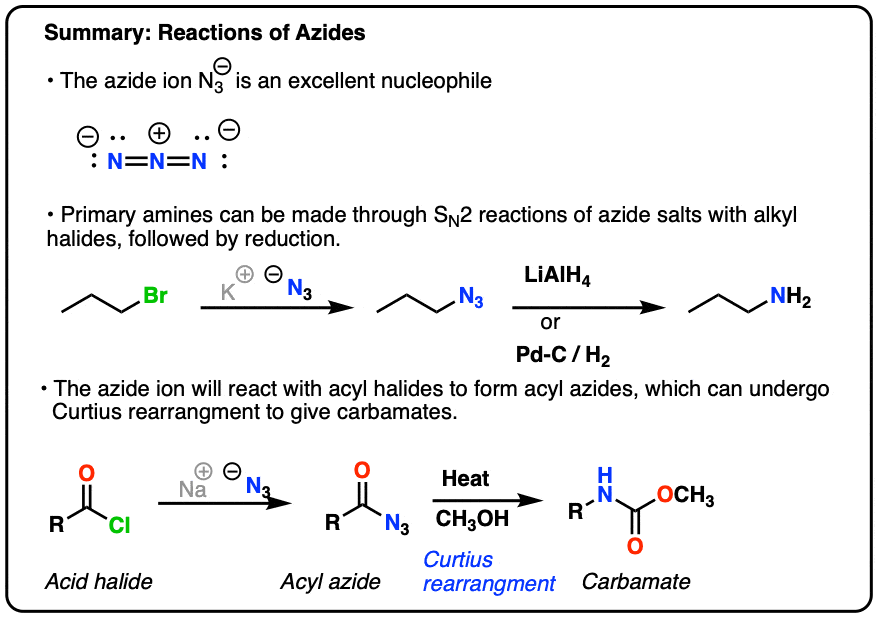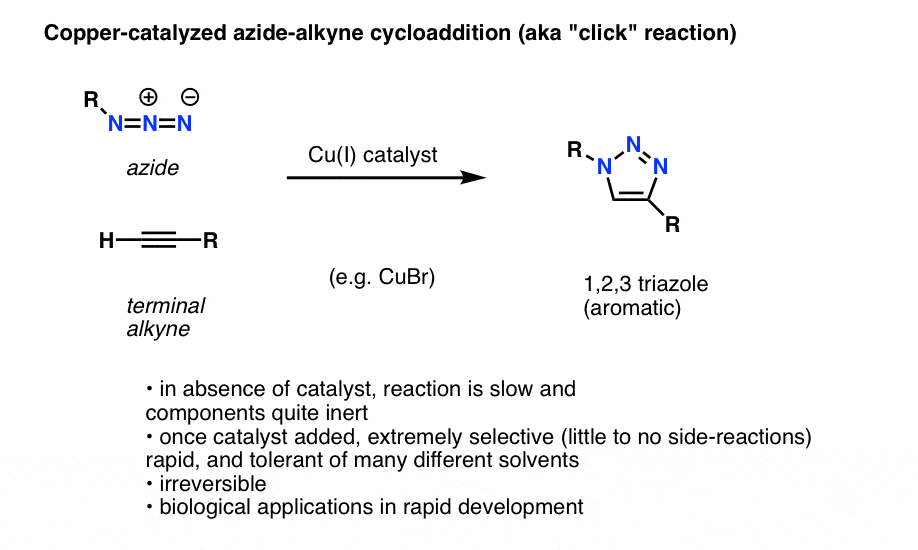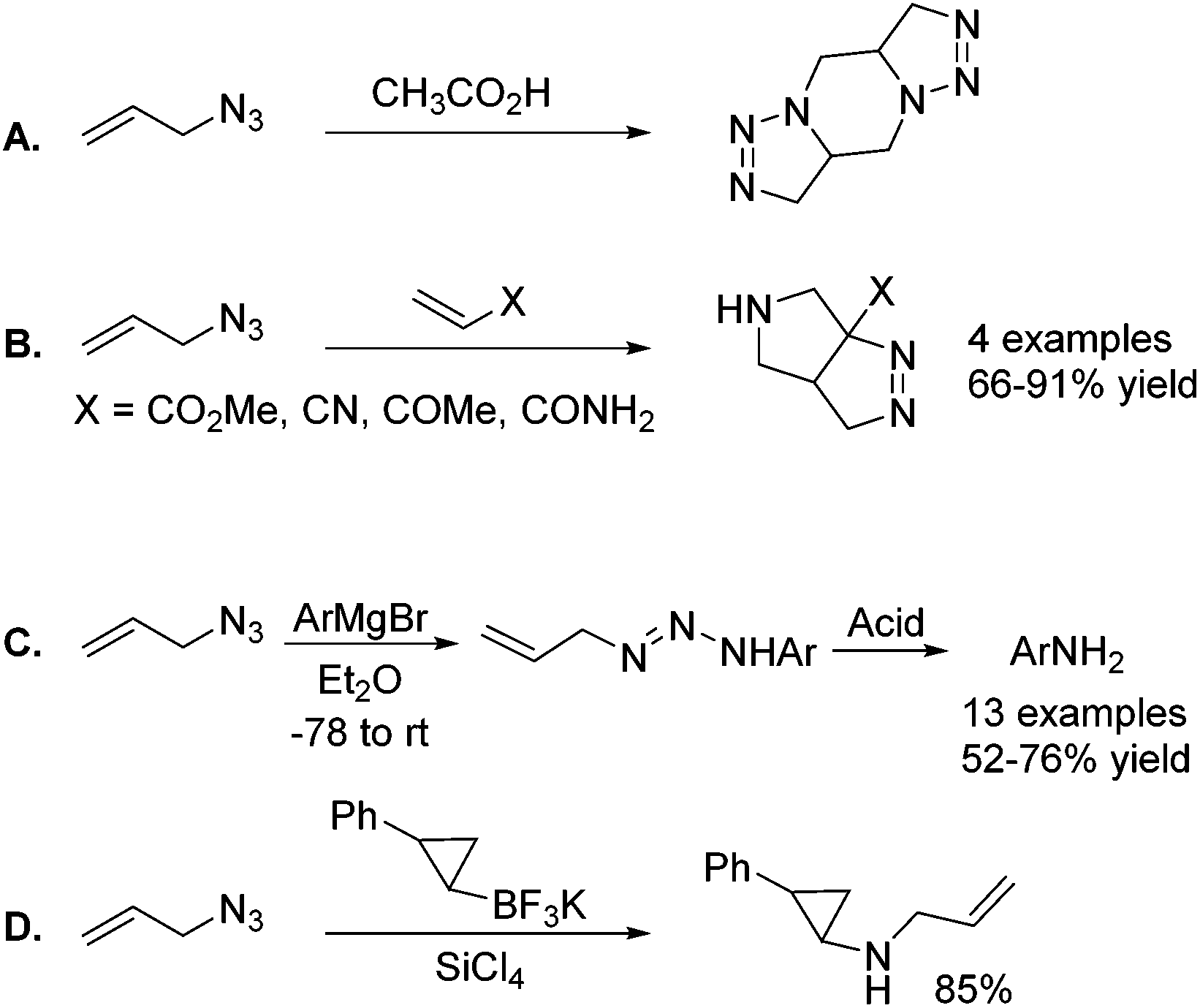
Allylic azides: synthesis, reactivity, and the Winstein rearrangement - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB03178A

Water/Alkali-Catalyzed Reactions of Azides with 2-Cyanothioacetamides. Eco-Friendly Synthesis of Monocyclic and Bicyclic 1,2,3-Thiadiazole-4-carbimidamides and 5-Amino-1,2,3-triazole-4-carbothioamides | The Journal of Organic Chemistry
![Molecules | Free Full-Text | SnAr Reactions of 2,4-Diazidopyrido[3,2-d]pyrimidine and Azide-Tetrazole Equilibrium Studies of the Obtained 5-Substituted Tetrazolo[1,5-a]pyrido[2,3-e]pyrimidines Molecules | Free Full-Text | SnAr Reactions of 2,4-Diazidopyrido[3,2-d]pyrimidine and Azide-Tetrazole Equilibrium Studies of the Obtained 5-Substituted Tetrazolo[1,5-a]pyrido[2,3-e]pyrimidines](https://www.mdpi.com/molecules/molecules-27-07675/article_deploy/html/images/molecules-27-07675-g001.png)
Molecules | Free Full-Text | SnAr Reactions of 2,4-Diazidopyrido[3,2-d]pyrimidine and Azide-Tetrazole Equilibrium Studies of the Obtained 5-Substituted Tetrazolo[1,5-a]pyrido[2,3-e]pyrimidines

Molecules | Free Full-Text | Reactive & Efficient: Organic Azides as Cross-Linkers in Material Sciences

Cu(I) Mediated Azidation of Halobenzenes, and Cu Catalysed Selective Azide Reduction to Corresponding Amines - Roemer - 2022 - Advanced Synthesis & Catalysis - Wiley Online Library
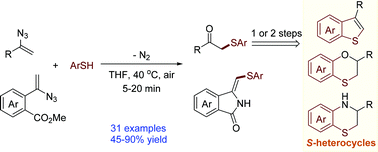
An aryl thiol–vinyl azide coupling reaction and a thiol–vinyl azide coupling/cyclization cascade: efficient synthesis of β-ketosulfides and arene-fused 5-methylene-2-pyrrolidinone derivatives - Organic & Biomolecular Chemistry (RSC Publishing)
![Azidation vs [3 + 2]-cycloaddition: Chemoselective reaction of sodium azide towards o-alkynylaldehydes for the synthesis of N-heterocycles - ScienceDirect Azidation vs [3 + 2]-cycloaddition: Chemoselective reaction of sodium azide towards o-alkynylaldehydes for the synthesis of N-heterocycles - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040403919305611-gr2.jpg)
Azidation vs [3 + 2]-cycloaddition: Chemoselective reaction of sodium azide towards o-alkynylaldehydes for the synthesis of N-heterocycles - ScienceDirect
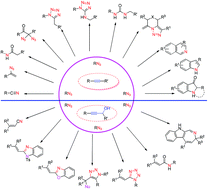
Recent advances in the tandem reaction of azides with alkynes or alkynols - Organic & Biomolecular Chemistry (RSC Publishing)
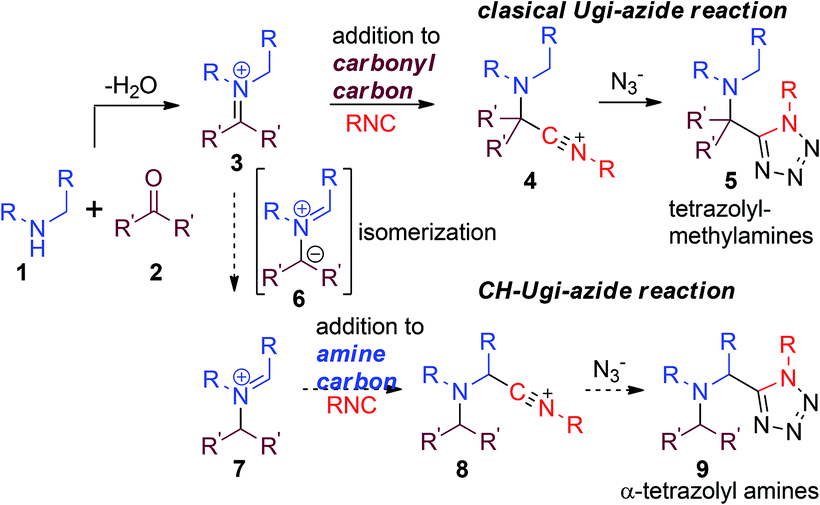
C–H functionalization enabled stereoselective Ugi-azide reaction to α-tetrazolyl alicyclic amines - Green Chemistry (RSC Publishing) DOI:10.1039/C8GC01544A